Gene silencing of NOB1 by lentivirus suppresses growth and migration of human osteosarcoma cells
- PMID: 24714960
- PMCID: PMC4055445
- DOI: 10.3892/mmr.2014.2119
Gene silencing of NOB1 by lentivirus suppresses growth and migration of human osteosarcoma cells
Erratum in
-
[Corrigendum] Gene silencing of NOB1 by lentivirus suppresses growth and migration of human osteosarcoma cells.Mol Med Rep. 2023 Feb;27(2):22. doi: 10.3892/mmr.2022.12909. Epub 2022 Dec 9. Mol Med Rep. 2023. PMID: 36484386 Free PMC article.
Abstract
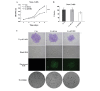
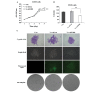
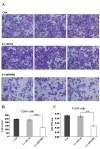
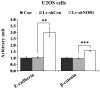
NIN1/RPN12 binding protein 1 homolog (Saccharomyces cerevisiae) (NOB1) encodes a chaperone protein that joins the 20S proteasome with the 19S regulatory particle in the nucleus and facilitates the biogenesis of the 26S proteasome, which plays a role in maintaining cellular homeostasis by controlling protein degradation. In order to investigate the role of NOB1 in osteosarcoma, NOB1 protein expression in human osteosarcoma cell lines was assessed using western blot analysis. Lentivirus-mediated short hairpin RNA was employed to knock down NOB1, and the effects of NOB1 silencing on cell growth were assessed using MTT, colony formation and cell cycle assays. Cell migration was observed using the Transwell assay. In addition, the expression levels of E-cadherin and β-catenin were examined by western blot analysis. Functional analysis indicated that NOB1-knockdown markedly inhibited cell growth and caused G2/M-phase arrest in human osteosarcoma cells. Furthermore, NOB1 inhibition decreased cell migration and increased E-cadherin and β-catenin expression in U2OS cells. In conclusion, the present study suggested that NOB1 depletion may inhibit osteosarcoma development by increasing E-cadherin and β-catenin expression and, for the first time, indicated the potential of NOB1 as a target in osteosarcoma treatment.
Figures


Similar articles
-
Knockdown of NOB1 expression by RNAi inhibits cellular proliferation and migration in human gliomas.Gene. 2013 Oct 10;528(2):146-53. doi: 10.1016/j.gene.2013.07.032. Epub 2013 Jul 31. Gene. 2013. PMID: 23911301
-
MiR-363 suppresses cell migration, invasion, and epithelial-mesenchymal transition of osteosarcoma by binding to NOB1.World J Surg Oncol. 2020 May 1;18(1):83. doi: 10.1186/s12957-020-01859-y. World J Surg Oncol. 2020. PMID: 32357945 Free PMC article.
-
Lentivirus-mediated gene silencing of NOB1 suppresses non-small cell lung cancer cell proliferation.Oncol Rep. 2015 Sep;34(3):1510-6. doi: 10.3892/or.2015.4132. Epub 2015 Jul 14. Oncol Rep. 2015. PMID: 26178254
-
NOB1: A Potential Biomarker or Target in Cancer.Curr Drug Targets. 2019;20(10):1081-1089. doi: 10.2174/1389450120666190308145346. Curr Drug Targets. 2019. PMID: 30854959 Review.
-
Ribosome Biogenesis and Cancer: Insights into NOB1 and PNO1 Mechanisms.Curr Pharm Des. 2024;30(37):2911-2921. doi: 10.2174/0113816128301870240730071910. Curr Pharm Des. 2024. PMID: 39143880 Review.
Cited by
-
A three gene-based risk score predicts prognosis of resected non-small-cell lung cancer.Int J Clin Exp Pathol. 2015 Dec 1;8(12):16081-8. eCollection 2015. Int J Clin Exp Pathol. 2015. PMID: 26884885 Free PMC article.
-
Regulation of Molecular Targets in Osteosarcoma Treatment.Int J Mol Sci. 2022 Oct 20;23(20):12583. doi: 10.3390/ijms232012583. Int J Mol Sci. 2022. PMID: 36293439 Free PMC article. Review.
-
Anticancer activity of NOB1-targeted shRNA combination with TRAIL in epithelial ovarian cancer cells.Int J Clin Exp Pathol. 2015 Sep 1;8(9):10061-71. eCollection 2015. Int J Clin Exp Pathol. 2015. Retraction in: Int J Clin Exp Pathol. 2021 Jul 15;14(7):856. PMID: 26617713 Free PMC article. Retracted.
-
hsa_circ0021347 as a Potential Target Regulated by B7-H3 in Modulating the Malignant Characteristics of Osteosarcoma.Biomed Res Int. 2019 Dec 17;2019:9301989. doi: 10.1155/2019/9301989. eCollection 2019. Biomed Res Int. 2019. PMID: 31950059 Free PMC article.
-
Review of LINC00707: A Novel LncRNA and Promising Biomarker for Human Diseases.Front Cell Dev Biol. 2022 Jan 26;10:813963. doi: 10.3389/fcell.2022.813963. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 35155429 Free PMC article. Review.
References
-
- Glickman MH, Ciechanover A. The ubiquitin-proteasome proteolytic pathway: destruction for the sake of construction. Physiol Rev. 2002;82:373–428. - PubMed
-
- Ferrell K, Wilkinson CR, Dubiel W, Gordon C. Regulatory subunit interactions of the 26S proteasome, a complex problem. Trends Biochem Sci. 2000;25:83–88. - PubMed
-
- Frezza M, Schmit S, Dou QP. Targeting the ubiquitin-proteasome pathway: an emerging concept in cancer therapy. Curr Top Med Chem. 2011;11:2888–2905. - PubMed
-
- Yerlikaya A, Yöntem M. The significance of ubiquitin proteasome pathway in cancer development. Recent Pat Anticancer Drug Discov. 2013;8:298–309. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

