Human enteroids as an ex-vivo model of host-pathogen interactions in the gastrointestinal tract
- PMID: 24719375
- PMCID: PMC4380516
- DOI: 10.1177/1535370214529398
Human enteroids as an ex-vivo model of host-pathogen interactions in the gastrointestinal tract
Abstract
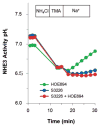
Currently, 9 out of 10 experimental drugs fail in clinical studies. This has caused a 40% plunge in the number of drugs approved by the US Food and Drug Administration (FDA) since 2005. It has been suggested that the mechanistic differences between human diseases modeled in animals (mostly rodents) and the pathophysiology of human diseases might be one of the critical factors that contribute to drug failure in clinical trials. Rapid progress in the field of human stem cell technology has allowed the in-vitro recreation of human tissue that should complement and expand upon the limitations of cell and animal models currently used to study human diseases and drug toxicity. Recent success in the identification and isolation of human intestinal epithelial stem cells (Lgr5(+)) from the small intestine and colon has led to culture of functional intestinal epithelial units termed organoids or enteroids. Intestinal enteroids are comprised of all four types of normal epithelial cells and develop a crypt-villus differentiation axis. They demonstrate major intestinal physiologic functions, including Na(+) absorption and Cl(-) secretion. This review discusses the recent progress in establishing human enteroids as a model of infectious diarrheal diseases such as cholera, rotavirus, and enterohemorrhagic Escherichia coli, and use of the enteroids to determine ways to correct the diarrhea-induced ion transport abnormalities via drug therapy.
Keywords: Enteroids; host–pathogen interaction; intestinal pathophysiology; intestinal physiology.
© 2014 by the Society for Experimental Biology and Medicine.
Figures


References
-
- Sato T, Vries RG, Snippert HJ, van de Wetering M, Barker N, Stange DE, van Es JH, Abo A, Kujala P, Peters PJ, Clevers H. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. 2009;459:262–5. - PubMed
-
- Jung P, Sato T, Merlos-Suárez A, Barriga FM, Iglesias M, Rossell D, Auer H, Gallardo M, Blasco MA, Sancho E, Clevers H, Batlle E. Isolation and in vitro expansion of human colonic stem cells. Nat Med. 2011;17:1225–7. - PubMed
-
- Sato T, Stange DE, Ferrante M, Vries RGJ, van Es JH, van den Brink S, van Houdt WJ, Pronk A, van Gorp J, Siersema PD, Clevers H. Long-term expansion of epithelial organoids from human colon, adenoma, adeno-carcinoma, and Barrett’s epithelium. Gastroenterology. 2011;141:1762–72. - PubMed
-
- Woodcock J, Woosley R. The FDA critical path initiative and its influence on new drug development. Annu Rev Med. 2008;59:1–12. - PubMed
-
- Hidalgo IJ, Raub TJ, Borchardt RT. Characterization of the human colon carcinoma cell line (Caco-2) as a model system for intestinal epithelial permeability. Gastroenterology. 1989;96:736–49. - PubMed
Publication types
MeSH terms
Grants and funding
- R24 DK099803/DK/NIDDK NIH HHS/United States
- R01-DK061765/DK/NIDDK NIH HHS/United States
- U19 AI116497/AI/NIAID NIH HHS/United States
- R01 DK026523/DK/NIDDK NIH HHS/United States
- P30 DK056338/DK/NIDDK NIH HHS/United States
- T32 DK007632/DK/NIDDK NIH HHS/United States
- K08 DK088950/DK/NIDDK NIH HHS/United States
- P01 DK072084/DK/NIDDK NIH HHS/United States
- P01-DK072084/DK/NIDDK NIH HHS/United States
- K01-DK088950/DK/NIDDK NIH HHS/United States
- R01-DK026523/DK/NIDDK NIH HHS/United States
- R24 DK064388/DK/NIDDK NIH HHS/United States
- K01 DK093657/DK/NIDDK NIH HHS/United States
- P30 DK089502/DK/NIDDK NIH HHS/United States
- R01 DK061765/DK/NIDDK NIH HHS/United States
- R01-AI080656/AI/NIAID NIH HHS/United States
- P30-DK089502/DK/NIDDK NIH HHS/United States
- R01 AI080656/AI/NIAID NIH HHS/United States
- U18 TR000552/TR/NCATS NIH HHS/United States
- U18-TR000552/TR/NCATS NIH HHS/United States
- P30-DK056338/DK/NIDDK NIH HHS/United States
- T32-DK007632/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

