Evidence for the use of isoflurane as a replacement for chloral hydrate anesthesia in experimental stroke: an ethical issue
- PMID: 24719888
- PMCID: PMC3955691
- DOI: 10.1155/2014/802539
Evidence for the use of isoflurane as a replacement for chloral hydrate anesthesia in experimental stroke: an ethical issue
Abstract
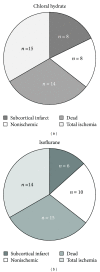
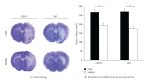
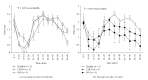
Since an ethical issue has been raised regarding the use of the well-known anesthetic agent chloral hydrate, owing to its mutagenic and carcinogenic effects in animals, attention of neuroscientists has turned to finding out an alternative agent able to meet not only potency, safety, and analgesic efficacy, but also reduced neuroprotective effect for stroke research. The aim of this study was to compare the potential of chloral hydrate and isoflurane for both modulating the action of the experimental neuroprotectant MK801 and exerting analgesia. After middle cerebral artery occlusion in rats, no difference was observed in 24 h survival rate, success of ischemia, or infarct volume reduction between both anesthetics. However, isoflurane exerted a more pronounced analgesic effect than chloral hydrate as evidenced by formalin test 3 hours after anesthesia onset, thus encouraging the use of isoflurane in experimental stroke models.
Figures


References
-
- Kao T-K, Ou Y-C, Liao S-L, et al. Opioids modulate post-ischemic progression in a rat model of stroke. Neurochemistry International. 2008;52(6):1256–1265. - PubMed
-
- Zhao P, Huang Y, Zuo Z. Opioid preconditioning induces opioid receptor-dependent delayed neuroprotection against ischemia in rats. Journal of Neuropathology and Experimental Neurology. 2006;65(10):945–952. - PubMed
-
- Gauillard J, Cheref S, VacheronTrystram M-N, Martin J-C. L'hydrate de chloral, un hypnotique à oublier? Mémoire Original. 2013;2:200–204.
-
- Özden S, Isenmann S. Neuroprotective properties of different anesthetics on axotomized rat retinal ganglion cells in vivo. Journal of Neurotrauma. 2004;21(1):73–82. - PubMed
-
- Flecknell P. Laboratory Animal Anesthesia. 2nd edition. San Diego, Calif, USA: Academic Press; 1996.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

