Hyperglycemia induces embryopathy, even in the absence of systemic maternal diabetes: an in vivo test of the fuel mediated teratogenesis hypothesis
- PMID: 24721120
- PMCID: PMC4067982
- DOI: 10.1016/j.reprotox.2014.03.013
Hyperglycemia induces embryopathy, even in the absence of systemic maternal diabetes: an in vivo test of the fuel mediated teratogenesis hypothesis
Abstract
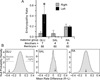
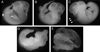
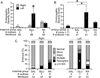
Embryonic exposure to excess circulating fuels is proposed to underlie diabetic embryopathy. To isolate the effects of hyperglycemia from the many systemic anomalies of diabetes, we infused 4 mg/min glucose into the left uterine artery of non-diabetic pregnant rats on gestation days (GD) 7-9. Right-sided embryos and dams exhibited no glucose elevation. Embryos were assessed on GD13, comparing the left versus right uterine horns. Hyperglycemic exposure increased rates of embryopathy, resorptions, and worsened embryopathy severity. By contrast, saline infusion did not affect any of these parameters. To assess for possible embryopathy susceptibility bias between uterine horns, separate dams were given retinoic acid (25mg/kg, a mildly embryopathic dose) systemically on GD7.5. The resultant embryopathy rates were equivalent between uterine horns. We conclude that hyperglycemia, even in the absence of systemic maternal diabetes, is sufficient to produce in vivo embryopathy during organogenesis.
Keywords: Animal model; Diabetes; Embryopathy; Fuel-mediated teratogenesis; Hyperglycemia; Teratogenesis.
Copyright © 2014 Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Figures


References
-
- Jensen DM, Damm P, Moelsted-Pedersen L, Ovesen P, Westergaard JG, Moeller M, et al. Outcomes in type 1 diabetic pregnancies: a nationwide, population-based study. Diabetes Care. 2004;27:2819–2823. - PubMed
-
- Bell R, Glinianaia SV, Tennant PWG, Bilous RW, Rankin J. Peri-conception hyperglycaemia and nephropathy are associated with risk of congenital anomaly in women with pre-existing diabetes: a population-based cohort study. Diabetologia. 2012 - PubMed
-
- Al-Agha R, Firth RG, Byrne M, Murray S, Daly S, Foley M, et al. Outcome of pregnancy in type 1 diabetes mellitus (T1DMP): results from combined diabetes-obstetrical clinics in Dublin in three university teaching hospitals (1995–2006) Ir J Med Sci. 2012;181:105–109. - PubMed
-
- Weintrob N, Karp M, Hod M. Short- and long-range complications in offspring of diabetic mothers. J Diabetes Complicat. 1996;10:294–301. - PubMed
-
- Reece EA, Homko CJ. Prepregnancy care and the prevention of fetal malformations in the pregnancy complicated by diabetes. Clin Obstet Gynecol. 2007;50:990–997. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

