Bevacizumab in treatment of high-risk ovarian cancer--a cost-effectiveness analysis
- PMID: 24721817
- PMCID: PMC4012959
- DOI: 10.1634/theoncologist.2013-0322
Bevacizumab in treatment of high-risk ovarian cancer--a cost-effectiveness analysis
Abstract
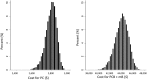
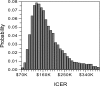
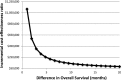
The objective of this study was to evaluate a cost-effectiveness strategy of bevacizumab in a subset of high-risk advanced ovarian cancer patients with survival benefit. Methods. A subset analysis of the International Collaboration on Ovarian Neoplasms 7 trial showed that additions of bevacizumab (B) and maintenance bevacizumab (mB) to paclitaxel (P) and carboplatin (C) improved the overall survival (OS) of high-risk advanced cancer patients. Actual and estimated costs of treatment were determined from Medicare payment. Incremental cost-effectiveness ratio per life-year saved was established. Results. The estimated cost of PC is $535 per cycle; PCB + mB (7.5 mg/kg) is $3,760 per cycle for the first 6 cycles and then $3,225 per cycle for 12 mB cycles. Of 465 high-risk stage IIIC (>1 cm residual) or stage IV patients, the previously reported OS after PC was 28.8 months versus 36.6 months in those who underwent PCB + mB. With an estimated 8-month improvement in OS, the incremental cost-effectiveness ratio of B was $167,771 per life-year saved. Conclusion. In this clinically relevant subset of women with high-risk advanced ovarian cancer with overall survival benefit after bevacizumab, our economic model suggests that the incremental cost of bevacizumab was approximately $170,000.
摘要
目的 本研究的目的是在一个获得生存益处的高危晚期卵巢癌患者亚组中,对贝伐单抗治疗的一项成本效益策略进行评估。
方法 对国际协作组卵巢肿瘤 7 试验进行的一项亚组分析表明,在紫杉醇 (P) 和卡铂 (C) 的基础上添加贝伐单抗 (B) 和延用贝伐单抗 (mB) 可延长高危晚期癌症患者的总生存期 (OS)。我们根据 Medicare 付款情况确定了实际治疗成本和估计治疗成本, 并确立了每拯救一个寿命年所对应的成本效益增量比值。
结果 PC 的估计成本为每周期 535 美元;PCB + mB (7.5 mg/kg) 的估计成本在前 6 个周期内为每周期 3,760 美元,在后 12 个 mB 周期内为每周期 3,225 美元。在 465 名高危 III 期(残留灶 >1 cm)或 IV 期患者中,既往报告的 PC 后 OS 为 28.8 个月,而那些接受 PCB + mB 之患者的 OS 则为 36.6 个月。在 OS 估计延长 8 个月的情况下,由 B 导致的成本效益增加比率为每拯救一个寿命年即增加 167,771 美元成本。
结论 在这个使用贝伐单抗后获得生存益处的临床重要高危晚期卵巢癌女性亚组中,我们的经济模型表明,由贝伐单抗导致的成本增加值约为 170,000 美元。The Oncologist 2014;19:523–527
Keywords: Bevacizumab; Cost-effectiveness analysis; Markov chain; Ovarian cancer.
Conflict of interest statement
Disclosures of potential conflicts of interest may be found at the end of this article.
Figures

References
-
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin. 2012;62:10–29. - PubMed
-
- Presta LG, Chen H, O’Connor SJ, et al. Humanization of an anti-vascular endothelial growth factor monoclonal antibody for the therapy of solid tumors and other disorders. Cancer Res. 1997;57:4593–4599. - PubMed
-
- Burger RA, Sill MW, Monk BJ, et al. Phase II trial of bevacizumab in persistent or recurrent epithelial ovarian cancer or primary peritoneal cancer: A Gynecologic Oncology Group Study. J Clin Oncol. 2007;25:5165–5171. - PubMed
-
- Cannistra SA, Matulonis UA, Penson RT, et al. Phase II study of bevacizumab in patients with platinum-resistant ovarian cancer or peritoneal serous cancer. J Clin Oncol. 2007;25:5180–5186. - PubMed
-
- Burger RA, Brady MF, Bookman MA, et al. Incorporation of bevacizumab in the primary treatment of ovarian cancer. N Engl J Med. 2011;365:2473–2483. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

