Colchicine-induced degeneration of the micronucleus during conjugation in Tetrahymena
- PMID: 24728958
- PMCID: PMC4021357
- DOI: 10.1242/bio.20147708
Colchicine-induced degeneration of the micronucleus during conjugation in Tetrahymena
Abstract
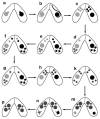
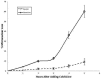
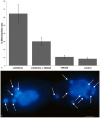
One of the most dramatic examples of nuclear morphogenesis occurs during conjugation in Tetrahymena when the micronucleus elongates to a size longer than the cell itself. After contraction to a spherical shape, the nucleus moves directly to chromosome separation in the first meiotic division. Here we investigate the consequences of interrupting the elongation process. Colchicine, a microtubule inhibitor, caused retraction of elongated structures. With time, cells began to lose their micronuclei, and by five hours more than half of the paired cells had at least one cell missing a micronucleus. After reversing the colchicine block, existing micronuclei did not undergo elongation again, nor did meiosis occur. These observations indicate that micronuclear elongation is critical to subsequent meiotic division. Further, nuclear elimination occurs, which could be due to meiotic failure or possibly a problem downstream from meiosis. An analysis of the process of colchicine-induced micronuclear degeneration indicated that it was regulated by a caspase-dependent mechanism, characteristic of apoptosis, and then resorbed by a lysosome-dependent autophagic mechanism. Amicronucleate cells failed to grow when returned to nutrient medium, likely because of a lesion in the post-conjugation reconstruction of a functioning oral apparatus. The ease by which a large number of nuclei are induced to "self-destruct" may make this system useful in investigating the link between colchicine treatment and nuclear death in Tetrahymena, and in investigating how nuclear death could be regulated in living cells more generally. Finally, we note that this phenomenon might relate to the evolution of amicronucleate species of Tetrahymena.
Keywords: Apoptosis; Autophagy; Ciliate; Meiosis; Micronucleus; Nuclear morphogenesis; Tetrahymena.
© 2014. Published by The Company of Biologists Ltd.
Conflict of interest statement
Figures





Similar articles
-
Cytofluorimetric analysis of nuclear DNA during meiosis, fertilization and macronuclear development in the ciliate Tetrahymena pyriformis, syngen 1.J Cell Sci. 1975 May;17(3):471-93. doi: 10.1242/jcs.17.3.471. J Cell Sci. 1975. PMID: 806605
-
Effect of the antitubulin drug nocodazole on meiosis and postmeiotic development in Tetrahymena thermophila. Induction of achiasmatic meiosis.Exp Cell Res. 1985 May;158(1):244-56. doi: 10.1016/0014-4827(85)90447-1. Exp Cell Res. 1985. PMID: 3996478
-
Histone rearrangements accompany nuclear differentiation and dedifferentiation in Tetrahymena.Dev Biol. 1984 Feb;101(2):282-94. doi: 10.1016/0012-1606(84)90142-8. Dev Biol. 1984. PMID: 6692982
-
Post-meiotic DNA synthesis in nocodazole-blocked nuclei during conjugation of Tetrahymena thermophila. Induction of polyploidy in the micronucleus.Exp Cell Res. 1986 Jun;164(2):562-7. doi: 10.1016/0014-4827(86)90055-8. Exp Cell Res. 1986. PMID: 3709684
-
Developmental progression of Tetrahymena through the cell cycle and conjugation.Methods Cell Biol. 2012;109:177-236. doi: 10.1016/B978-0-12-385967-9.00007-4. Methods Cell Biol. 2012. PMID: 22444146 Review.
Cited by
-
Evaluation of genotoxic potential of peptides used in nuclear medicine (PSMA -617 and -11, and ubiquicidine 29-41) using a flow-cytometric, semi-automated analysis of micronuclei frequency in cell cultures.Toxicol Rep. 2020 Feb 7;7:304-316. doi: 10.1016/j.toxrep.2020.02.003. eCollection 2020. Toxicol Rep. 2020. PMID: 32071884 Free PMC article.
-
A Novel 3D Scaffold for Cell Growth to Asses Electroporation Efficacy.Cells. 2019 Nov 19;8(11):1470. doi: 10.3390/cells8111470. Cells. 2019. PMID: 31752448 Free PMC article.
References
-
- Allen D. L., Linderman J. K., Roy R. R., Bigbee A. J., Grindeland R. E., Mukku V., Edgerton V. R. (1997). Apoptosis: a mechanism contributing to remodeling of skeletal muscle in response to hindlimb unweighting. Am. J. Physiol. 273, C579–C587. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

