Class I HDACs Affect DNA Replication, Repair, and Chromatin Structure: Implications for Cancer Therapy
- PMID: 24730655
- PMCID: PMC4492608
- DOI: 10.1089/ars.2014.5915
Class I HDACs Affect DNA Replication, Repair, and Chromatin Structure: Implications for Cancer Therapy
Abstract
Significance: The contribution of epigenetic alterations to cancer development and progression is becoming increasingly clear, prompting the development of epigenetic therapies. Histone deacetylase inhibitors (HDIs) represent one of the first classes of such therapy. Two HDIs, Vorinostat and Romidepsin, are broad-spectrum inhibitors that target multiple histone deacetylases (HDACs) and are FDA approved for the treatment of cutaneous T-cell lymphoma. However, the mechanism of action and the basis for the cancer-selective effects of these inhibitors are still unclear.
Recent advances: While the anti-tumor effects of HDIs have traditionally been attributed to their ability to modify gene expression after the accumulation of histone acetylation, recent studies have identified the effects of HDACs on DNA replication, DNA repair, and genome stability. In addition, the HDIs available in the clinic target multiple HDACs, making it difficult to assign either their anti-tumor effects or their associated toxicities to the inhibition of a single protein. However, recent studies in mouse models provide insights into the tissue-specific functions of individual HDACs and their involvement in mediating the effects of HDI therapy.
Critical issues: Here, we describe how altered replication contributes to the efficacy of HDAC-targeted therapies as well as discuss what knowledge mouse models have provided to our understanding of the specific functions of class I HDACs, their potential involvement in tumorigenesis, and how their disruption may contribute to toxicities associated with HDI treatment.
Future directions: Impairment of DNA replication by HDIs has important therapeutic implications. Future studies should assess how best to exploit these findings for therapeutic gain.
Figures

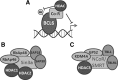
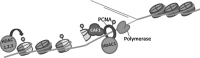

References
-
- Aggarwal BD. and Calvi BR. Chromatin regulates origin activity in Drosophila follicle cells. Nature 430: 372–376, 2004 - PubMed
-
- Ahmad A, Takami Y, and Nakayama T. WD repeats of the p48 subunit of chicken chromatin assembly factor-1 required for in vitro interaction with chicken histone deacetylase-2. J Biol Chem 274: 16646–16653, 1999 - PubMed
-
- Alabert C. and Groth A. Chromatin replication and epigenome maintenance. Nat Rev Mol Cell Biol 13: 153–167, 2012 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

