Linked production of pyroglutamate-modified proteins via self-cleavage of fusion tags with TEV protease and autonomous N-terminal cyclization with glutaminyl cyclase in vivo
- PMID: 24733552
- PMCID: PMC3986218
- DOI: 10.1371/journal.pone.0094812
Linked production of pyroglutamate-modified proteins via self-cleavage of fusion tags with TEV protease and autonomous N-terminal cyclization with glutaminyl cyclase in vivo
Abstract
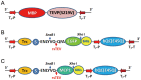
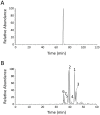
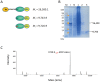
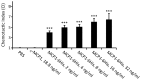
Overproduction of N-terminal pyroglutamate (pGlu)-modified proteins utilizing Escherichia coli or eukaryotic cells is a challenging work owing to the fact that the recombinant proteins need to be recovered by proteolytic removal of fusion tags to expose the N-terminal glutaminyl or glutamyl residue, which is then converted into pGlu catalyzed by the enzyme glutaminyl cyclase. Herein we describe a new method for production of N-terminal pGlu-containing proteins in vivo via intracellular self-cleavage of fusion tags by tobacco etch virus (TEV) protease and then immediate N-terminal cyclization of passenger target proteins by a bacterial glutaminyl cyclase. To combine with the sticky-end PCR cloning strategy, this design allows the gene of target proteins to be efficiently inserted into the expression vector using two unique cloning sites (i.e., SnaB I and Xho I), and the soluble and N-terminal pGlu-containing proteins are then produced in vivo. Our method has been successfully applied to the production of pGlu-modified enhanced green fluorescence protein and monocyte chemoattractant proteins. This design will facilitate the production of protein drugs and drug target proteins that possess an N-terminal pGlu residue required for their physiological activities.
Conflict of interest statement
Figures

Similar articles
-
Functional comparison of two human monocyte chemotactic protein-2 isoforms, role of the amino-terminal pyroglutamic acid and processing by CD26/dipeptidyl peptidase IV.Biochemistry. 1998 Sep 8;37(36):12672-80. doi: 10.1021/bi980497d. Biochemistry. 1998. PMID: 9730840
-
Phosphate ions and glutaminyl cyclases catalyze the cyclization of glutaminyl residues by facilitating synchronized proton transfers.Bioorg Chem. 2015 Jun;60:98-101. doi: 10.1016/j.bioorg.2015.04.005. Epub 2015 Apr 24. Bioorg Chem. 2015. PMID: 25981125
-
Inhibition of glutaminyl cyclase prevents pGlu-Abeta formation after intracortical/hippocampal microinjection in vivo/in situ.J Neurochem. 2008 Aug;106(3):1225-36. doi: 10.1111/j.1471-4159.2008.05471.x. Epub 2008 Jul 8. J Neurochem. 2008. PMID: 18627432
-
Human glutaminyl cyclase: Structure, function, inhibitors and involvement in Alzheimer's disease.Pharmacol Res. 2019 Sep;147:104342. doi: 10.1016/j.phrs.2019.104342. Epub 2019 Jul 6. Pharmacol Res. 2019. PMID: 31288079 Review.
-
Glutaminyl cyclases, the potential targets of cancer and neurodegenerative diseases.Eur J Pharmacol. 2022 Sep 15;931:175178. doi: 10.1016/j.ejphar.2022.175178. Epub 2022 Aug 7. Eur J Pharmacol. 2022. PMID: 35948163 Review.
Cited by
-
Metal Ion Binding to Human Glutaminyl Cyclase: A Structural Perspective.Int J Mol Sci. 2024 Jul 29;25(15):8279. doi: 10.3390/ijms25158279. Int J Mol Sci. 2024. PMID: 39125848 Free PMC article.
-
Identification of Glutaminyl Cyclase Genes Involved in Pyroglutamate Modification of Fungal Lignocellulolytic Enzymes.mBio. 2017 Jan 17;8(1):e02231-16. doi: 10.1128/mBio.02231-16. mBio. 2017. PMID: 28096492 Free PMC article.
-
Diverse protein manipulations with genetically encoded glutamic acid benzyl ester.Chem Sci. 2021 Jun 17;12(28):9778-9785. doi: 10.1039/d1sc01882e. eCollection 2021 Jul 21. Chem Sci. 2021. PMID: 34349951 Free PMC article.
-
Demethionylation of Pro-1 variants of 4-oxalocrotonate tautomerase in Escherichia coli by co-expression with an engineered methionine aminopeptidase.FEBS Open Bio. 2014 Jul 9;4:651-8. doi: 10.1016/j.fob.2014.07.003. eCollection 2014. FEBS Open Bio. 2014. PMID: 25161874 Free PMC article.
References
-
- Van Coillie E, Proost P, Van Aelst I, Struyf S, Polfliet M, et al. (1998) Functional comparison of two human monocyte chemotactic protein-2 isoforms, role of the amino-terminal pyroglutamic acid and processing by CD26/dipeptidyl peptidase IV. Biochemistry 37: 12672–12680. - PubMed
-
- Goren HJ, Bauce LG, Vale W (1977) Forces and structural limitations of binding of thyrotrophin-releasing factor to the thyrotrophin-releasing receptor: the pyroglutamic acid moiety. Mol Pharmacol 13: 606–614. - PubMed
-
- Kitabgi P, De Nadai F, Rovère C, Bidard JN (1992) Biosynthesis, maturation, release, and degradation of neurotensin and neuromedin N. Ann N Y Acad Sci 668: 30–42. - PubMed
-
- Smith AM, Watson SA (2000) Review article: gastrin and colorectal cancer. Aliment Pharmacol Ther 14: 1231–1247. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

