Glucocorticoid receptor status is a principal determinant of variability in the sensitivity of non-small-cell lung cancer cells to pemetrexed
- PMID: 24736075
- PMCID: PMC4075060
- DOI: 10.1097/JTO.0000000000000111
Glucocorticoid receptor status is a principal determinant of variability in the sensitivity of non-small-cell lung cancer cells to pemetrexed
Abstract
Introduction: Pemetrexed is an S-phase targeted drug in front-line or maintenance therapy of advanced nonsquamous non-small-cell lung cancer (NSCLC) but methods are needed for predicting the drug response. Dexamethasone is typically administered the day before, the day of, and the day after pemetrexed. As dexamethasone strongly regulates many genes including p53 through the glucocorticoid receptor (GR), we hypothesized that dexamethasone influences tumor response to pemetrexed.
Methods: Eight nonsquamous NSCLC cell line models with varied p53 and GRα/GRβ status were used for gene expression and cell-cycle analyses and for loss- or gain-of-function experiments.
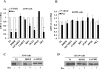
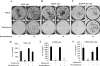
Results: In three cell lines dexamethasone profoundly, but reversibly, suppressed the fraction of S-phase cells. Dexamethasone also reversibly repressed expression of thymidylate synthase and dihydrofolate reductase, which are primary targets of pemetrexed but are also quintessential S-phase enzymes as well as the S-phase-dependent expression of thymidine kinase 1. Dexamethasone also decreased expression of the major pemetrexed transporters, the reduced folate carrier and the proton coupled folate transporter. Only cells expressing relatively high GRα showed these dexamethasone effects, regardless of p53 status. In cells expressing low GRα, the dexamethasone response was rescued by ectopic GRα. Further, depletion of p53 did not attenuate the dexamethasone effects. The presence of dexamethasone during pemetrexed treatment protected against pemetrexed cytotoxicity in only the dexamethasone responsive cells.
Conclusions: The results predict that in nonsquamous NSCLC tumors, reversible S-phase suppression by dexamethasone, possibly combined with a reduction in the drug transporters, attenuates responsiveness to pemetrexed and that GR status is a principal determinant of tumor variability of this response.
Figures

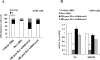
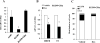
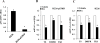
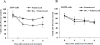
Similar articles
-
Significance of glucocorticoid receptor expression in patients with non-small cell lung cancer treated with pemetrexed-based chemotherapy.Cancer Chemother Pharmacol. 2017 Oct;80(4):851-860. doi: 10.1007/s00280-017-3399-1. Epub 2017 Jul 28. Cancer Chemother Pharmacol. 2017. PMID: 28755014
-
Genetic determinants of pemetrexed responsiveness and nonresponsiveness in non-small cell lung cancer cells.J Thorac Oncol. 2010 Aug;5(8):1143-51. doi: 10.1097/JTO.0b013e3181e0b954. J Thorac Oncol. 2010. PMID: 20559153
-
Phase II study of pemetrexed and cisplatin plus cetuximab followed by pemetrexed and cetuximab maintenance therapy in patients with advanced nonsquamous non-small cell lung cancer.Lung Cancer. 2013 Sep;81(3):428-434. doi: 10.1016/j.lungcan.2013.05.010. Epub 2013 Jun 20. Lung Cancer. 2013. PMID: 23790468 Clinical Trial.
-
Pemetrexed combination therapy in the treatment of non-small cell lung cancer.Semin Oncol. 2002 Apr;29(2 Suppl 5):23-9. doi: 10.1053/sonc.2002.30768. Semin Oncol. 2002. PMID: 12023789 Review.
-
Pharmacological aspects of the enzastaurin-pemetrexed combination in non-small cell lung cancer (NSCLC).Curr Drug Targets. 2010 Jan;11(1):12-28. doi: 10.2174/138945010790031009. Curr Drug Targets. 2010. PMID: 19839931 Review.
Cited by
-
Chronic p27Kip1 Induction by Dexamethasone Causes Senescence Phenotype and Permanent Cell Cycle Blockade in Lung Adenocarcinoma Cells Over-expressing Glucocorticoid Receptor.Sci Rep. 2018 Oct 30;8(1):16006. doi: 10.1038/s41598-018-34475-8. Sci Rep. 2018. PMID: 30375484 Free PMC article.
-
Synthetic Lethality through the Lens of Medicinal Chemistry.J Med Chem. 2020 Dec 10;63(23):14151-14183. doi: 10.1021/acs.jmedchem.0c00766. Epub 2020 Nov 2. J Med Chem. 2020. PMID: 33135887 Free PMC article. Review.
-
Senescence-associated secretory proteins induced in lung adenocarcinoma by extended treatment with dexamethasone enhance migration and activation of lymphocytes.Cancer Immunol Immunother. 2023 May;72(5):1273-1284. doi: 10.1007/s00262-022-03332-z. Epub 2022 Nov 25. Cancer Immunol Immunother. 2023. PMID: 36434273 Free PMC article.
-
Disease- and treatment-associated acquired glucocorticoid resistance.Endocr Connect. 2018 Dec;7(12):R328-R349. doi: 10.1530/EC-18-0421. Endocr Connect. 2018. PMID: 30352419 Free PMC article. Review.
-
Regulation of differential proton-coupled folate transporter gene expression in human tumors: transactivation by KLF15 with NRF-1 and the role of Sp1.Biochem J. 2019 Apr 26;476(8):1247-1266. doi: 10.1042/BCJ20180394. Biochem J. 2019. PMID: 30914440 Free PMC article.
References
-
- Ciuleanu T, et al. Maintenance pemetrexed plus best supportive care versus placebo plus best supportive care for non-small-cell lung cancer: a randomised, double-blind, phase 3 study. Lancet. 2009;374(9699):1432–40. - PubMed
-
- Hanna N, et al. Randomized phase III trial of pemetrexed versus docetaxel in patients with non-small-cell lung cancer previously treated with chemotherapy. J Clin Oncol. 2004;22(9):1589–97. - PubMed
-
- Scagliotti G, et al. The differential efficacy of pemetrexed according to NSCLC histology: a review of two Phase III studies. Oncologist. 2009;14(3):253–63. - PubMed
-
- Scagliotti GV, et al. Phase III study comparing cisplatin plus gemcitabine with cisplatin plus pemetrexed in chemotherapy-naive patients with advanced-stage non-small-cell lung cancer. J Clin Oncol. 2008;26(21):3543–51. - PubMed
-
- Chattopadhyay S, Moran RG, Goldman ID. Pemetrexed: biochemical and cellular pharmacology, mechanisms, and clinical applications. Mol Cancer Ther. 2007;6(2):404–17. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

