p53 modulates Hsp90 ATPase activity and regulates aryl hydrocarbon receptor signaling
- PMID: 24736433
- PMCID: PMC4074578
- DOI: 10.1158/1940-6207.CAPR-14-0051
p53 modulates Hsp90 ATPase activity and regulates aryl hydrocarbon receptor signaling
Retraction in
-
Retraction: p53 Modulates Hsp90 ATPase Activity and Regulates Aryl Hydrocarbon Receptor Signaling.Cancer Prev Res (Phila). 2022 Jun 2;15(6):408. doi: 10.1158/1940-6207.CAPR-22-0207. Cancer Prev Res (Phila). 2022. PMID: 35652230 Free PMC article. No abstract available.
Abstract
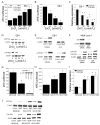
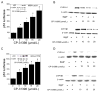
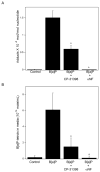
The aryl hydrocarbon receptor (AhR), a client protein of heat shock protein 90 (Hsp90), is a ligand-activated transcription factor that plays a role in polycyclic aromatic hydrocarbon (PAH)-induced carcinogenesis. Tobacco smoke activates AhR signaling leading to increased transcription of CYP1A1 and CYP1B1, which encode proteins that convert PAHs to mutagens. Recently, p53 was found to regulate Hsp90 ATPase activity via effects on activator of Hsp90 ATPase (Aha1). It is possible, therefore, that AhR-dependent expression of CYP1A1 and CYP1B1 might be affected by p53 status. The main objective of this study was to determine whether p53 modulated AhR-dependent gene expression and PAH metabolism. Here, we show that silencing p53 led to elevated Aha1 levels, increased Hsp90 ATPase activity, and enhanced CYP1A1 and CYP1B1 expression. Overexpression of wild-type p53 suppressed levels of CYP1A1 and CYP1B1. The significance of Aha1 in mediating these p53-dependent effects was determined. Silencing of Aha1 led to reduced Hsp90 ATPase activity and downregulation of CYP1A1 and CYP1B1. In contrast, overexpressing Aha1 was associated with increased Hsp90 ATPase activity and elevated levels of CYP1A1 and CYP1B1. Using p53 heterozygous mutant epithelial cells from patients with Li-Fraumeni syndrome, we show that monoallelic mutation of p53 was associated with elevated levels of CYP1A1 and CYP1B1 under both basal conditions and following treatment with benzo[a]pyrene. Treatment with CP-31398, a p53 rescue compound, suppressed benzo[a]pyrene-mediated induction of CYP1A1 and CYP1B1 and the formation of DNA adducts. Collectively, our results suggest that p53 affects AhR-dependent gene expression, PAH metabolism, and possibly carcinogenesis.
©2014 American Association for Cancer Research.
Conflict of interest statement
The authors have no conflicts of interest to declare
Figures



Comment in
-
Findings of Research Misconduct.Fed Regist. 2023 Sep 13;88(176):62800-62803. Fed Regist. 2023. PMID: 37736072 Free PMC article. No abstract available.
-
Findings of Research Misconduct.Fed Regist. 2023 Sep 13;88(176):62803-62807. Fed Regist. 2023. PMID: 37736073 Free PMC article. No abstract available.
References
-
- Kasai A, Hiramatsu N, Hayakawa K, Yao J, Maeda S, Kitamura M. High levels of dioxin-like potential in cigarette smoke evidenced by in vitro and in vivo biosensing. Cancer Res. 2006;66:7143–50. - PubMed
-
- Moennikes O, Loeppen S, Buchmann A, Andersson P, Ittrich C, Poellinger L, et al. A constitutively active dioxin/aryl hydrocarbon receptor promotes hepatocarcinogenesis in mice. Cancer Res. 2004;64:4707–10. - PubMed
-
- Port JL, Yamaguchi K, Du B, De Lorenzo M, Chang M, Heerdt PM, et al. Tobacco smoke induces CYP1B1 in the aerodigestive tract. Carcinogenesis. 2004;25:2275–81. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

