Nitroheterocyclic compounds are more efficacious than CYP51 inhibitors against Trypanosoma cruzi: implications for Chagas disease drug discovery and development
- PMID: 24736467
- PMCID: PMC4004771
- DOI: 10.1038/srep04703
Nitroheterocyclic compounds are more efficacious than CYP51 inhibitors against Trypanosoma cruzi: implications for Chagas disease drug discovery and development
Abstract
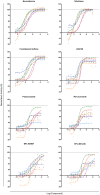
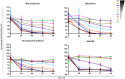
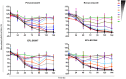
Advocacy for better drugs and access to treatment has boosted the interest in drug discovery and development for Chagas disease, a chronic infection caused by the genetically heterogeneous parasite, Trypanosoma cruzi. In this work new in vitro assays were used to gain a better understanding of the antitrypanosomal properties of the most advanced antichagasic lead and clinical compounds, the nitroheterocyclics benznidazole, nifurtimox and fexinidazole sulfone, the oxaborole AN4169, and four ergosterol biosynthesis inhibitors--posaconazole, ravuconazole, EPL-BS967 and EPL-BS1246. Two types of assays were developed: one for evaluation of potency and efficacy in dose-response against a panel of T. cruzi stocks representing all current discrete typing units (DTUs), and a time-kill assay. Although less potent, the nitroheterocyclics and the oxaborole showed broad efficacy against all T. cruzi tested and were rapidly trypanocidal, whilst ergosterol biosynthesis inhibitors showed variable activity that was both compound- and strain-specific, and were unable to eradicate intracellular infection even after 7 days of continuous compound exposure at most efficacious concentrations. These findings contest previous reports of variable responses to nitroderivatives among different T. cruzi strains and further challenge the introduction of ergosterol biosynthesis inhibitors as new single chemotherapeutic agents for the treatment of Chagas disease.
Figures
References
-
- WHO. Working to overcome the global impact of neglected tropical diseases. Available from http://www.who.int/neglected_diseases/2010report/en/ (2010) (Accessed on 18/02/2014).
-
- Gascon J., Bern C. & Pinazo M.-J. Chagas disease in Spain, the United States and other non-endemic countries. Acta Trop. 115, 22–27 (2010). - PubMed
-
- Bern C. & Montgomery S. P. An estimate of the burden of Chagas disease in the United States. Clin. Infec. Dis. 49, e52–4 (2009). - PubMed
-
- Rassi A. J., Rassi A. & Marcondes de Rezende J. American trypanosomiasis (Chagas disease). Infect. Dis. Clin. North Am. 26, 275–291 (2012). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

