Peptide transporter isoforms are discriminated by the fluorophore-conjugated dipeptides β-Ala- and d-Ala-Lys-N-7-amino-4-methylcoumarin-3-acetic acid
- PMID: 24744852
- PMCID: PMC3970736
- DOI: 10.1002/phy2.165
Peptide transporter isoforms are discriminated by the fluorophore-conjugated dipeptides β-Ala- and d-Ala-Lys-N-7-amino-4-methylcoumarin-3-acetic acid
Abstract
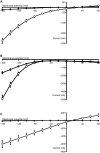
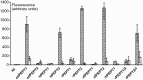
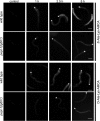
Peptide transporters of the SLC15 family are classified by structure and function into PEPT1 (low-affinity/high-capacity) and PEPT2 (high-affinity/low-capacity) isoforms. Despite the differences in kinetics, both transporter isoforms are reckoned to transport essentially all possible di- and tripeptides. We here report that the fluorophore-conjugated dipeptide derivatives β-Ala-Lys-N-7-amino-4-methylcoumarin-3-acetic acid (β-AK-AMCA) and d-Ala-Lys-N-7-amino-4-methylcoumarin-3-acetic acid (d-AK-AMCA) are transported by distinct PEPT isoforms in a species-specific manner. Transport of the fluorophore peptides was studied (1) in vitro after heterologous expression in Xenopus oocytes of PEPT1 and PEPT2 isoforms from different vertebrate species and of PEPT1 and PEPT2 transporters from Caenorhabditis elegans by using electrophysiological and fluorescence methods and (2) in vivo in C. elegans by using fluorescence methods. Our results indicate that both substrates are transported by the vertebrate "renal-type" and the C. elegans "intestinal-type" peptide transporter only. A systematic analysis among species finds four predicted amino acid residues along the sequence that may account for the substrate uptake differences observed between the vertebrate PEPT1/nematode PEPT2 and the vertebrate PEPT2/nematode PEPT1 subtype. This selectivity on basis of isoforms and species may be helpful in better defining the structure-function determinants of the proteins of the SLC15 family.
Keywords: Caenorhabditis elegans; PEPT1; PEPT2; di‐ and tripeptide; substrate specificity; transporter.
Figures

References
-
- Abe H., Satoh M., Miyauchi S., Shuto S., Matsuda A., Kamo N. 1999. Conjugation of dipeptide to fluorescent dyes enhances its affinity for a dipeptide transporter (PEPT1) in human intestinal Caco‐2 cells. Bioconjug. Chem.; 10:24-31 - PubMed
-
- Bednarczyk D., Mash E. A., Aavula B. R., Wright S. H. 2000. NBD‐TMA: a novel fluorescent substrate of the peritubular organic cation transporter of renal proximal tubules. Pflugers Arch.; 440:184-192 - PubMed
-
- Biegel A., Gebauer S., Hartrodt B., Brandsch M., Neubert K., Thondorf I. 2005. Three‐dimensional quantitative structure‐activity relationship analyses of beta‐lactam antibiotics and tripeptides as substrates of the mammalian H+/peptide cotransporter PEPT1. J. Med. Chem.; 48:4410-4419 - PubMed
-
- Biegel A., Gebauer S., Brandsch M., Neubert K., Thondorf I. 2006. Structural requirements for the substrates of the H+/peptide cotransporter PEPT2 determined by three‐dimensional quantitative structure‐activity relationship analysis. J. Med. Chem.; 49:4286-4296 - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

