Reduction of Na/K-ATPase affects cardiac remodeling and increases c-kit cell abundance in partial nephrectomized mice
- PMID: 24748592
- PMCID: PMC4059984
- DOI: 10.1152/ajpheart.00102.2014
Reduction of Na/K-ATPase affects cardiac remodeling and increases c-kit cell abundance in partial nephrectomized mice
Abstract
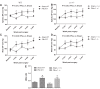
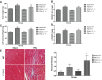
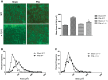
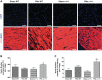
The current study examined the role of Na/K-ATPase α1-subunit in animals subjected to 5/6th partial nephrectomy (PNx) using Na/K-ATPase α1-heterozygous (α1(+/-)) mice and their wild-type (WT) littermates. After PNx, both WT and α1(+/-) animals displayed diastolic dimension increases, increased blood pressure, and increased cardiac hypertrophy. However, in the α1(+/-) animals we detected significant increases in cardiac cell death in PNx animals. Given that reduction of α1 elicited increased cardiac cell death with PNx, while at the same time these animals developed cardiac hypertrophy, an examination of cardiac cell number, and proliferative capabilities of those cells was carried out. Cardiac tissues were probed for the progenitor cell marker c-kit and the proliferation marker ki-67. The results revealed that α1(+/-) mice had significantly higher numbers of c-kit-positive and ki-67-positive cells, especially in the PNx group. We also found that α1(+/-) mice express higher levels of stem cell factor, a c-kit ligand, in their heart tissue and had higher circulating levels of stem cell factor than WT animals. In addition, PNx induced significant enlargement of cardiac myocytes in WT mice but has much less effect in α1(+/-) mice. However, the total cell number determined by nuclear counting is higher in α1(+/-) mice with PNx compared with WT mice. We conclude that PNx induces hypertrophic growth and high blood pressure regardless of Na/K-ATPase content change. However, total cardiac cell number as well as c-kit-positive cell number is increased in α1(+/-) mice with PNx.
Keywords: ATPases; apoptosis; cardiac hypertrophy; cardiac progenitor cells; cardiotonic steroids; caspase; cell proliferation; stem cells; uremic cardiomyopathy.
Copyright © 2014 the American Physiological Society.
Figures





Similar articles
-
Targeting Na/K-ATPase Signaling: A New Approach to Control Oxidative Stress.Curr Pharm Des. 2018;24(3):359-364. doi: 10.2174/1381612824666180110101052. Curr Pharm Des. 2018. PMID: 29318961 Free PMC article. Review.
-
Reduction of Na/K-ATPase potentiates marinobufagenin-induced cardiac dysfunction and myocyte apoptosis.J Biol Chem. 2012 May 11;287(20):16390-8. doi: 10.1074/jbc.M111.304451. Epub 2012 Mar 27. J Biol Chem. 2012. PMID: 22451662 Free PMC article.
-
Na/K-ATPase signaling mediates miR-29b-3p regulation and cardiac fibrosis formation in mice with chronic kidney disease.PLoS One. 2018 May 18;13(5):e0197688. doi: 10.1371/journal.pone.0197688. eCollection 2018. PLoS One. 2018. PMID: 29775473 Free PMC article.
-
Cardiac Oxidative Signaling and Physiological Hypertrophy in the Na/K-ATPase α1s/sα2s/s Mouse Model of High Affinity for Cardiotonic Steroids.Int J Mol Sci. 2021 Mar 27;22(7):3462. doi: 10.3390/ijms22073462. Int J Mol Sci. 2021. PMID: 33801629 Free PMC article.
-
The Redox-Sensitive Na/K-ATPase Signaling in Uremic Cardiomyopathy.Int J Mol Sci. 2020 Feb 13;21(4):1256. doi: 10.3390/ijms21041256. Int J Mol Sci. 2020. PMID: 32069992 Free PMC article. Review.
Cited by
-
Characterization of a Long Non-Coding RNA, the Antisense RNA of Na/K-ATPase α1 in Human Kidney Cells.Int J Mol Sci. 2018 Jul 21;19(7):2123. doi: 10.3390/ijms19072123. Int J Mol Sci. 2018. PMID: 30037072 Free PMC article.
-
The Effect of Electronic-Cigarette Vaping on Cardiac Function and Angiogenesis in Mice.Sci Rep. 2019 Mar 11;9(1):4085. doi: 10.1038/s41598-019-40847-5. Sci Rep. 2019. PMID: 30858470 Free PMC article.
-
Chronic inhalation of e-cigarette vapor containing nicotine disrupts airway barrier function and induces systemic inflammation and multiorgan fibrosis in mice.Am J Physiol Regul Integr Comp Physiol. 2018 Jun 1;314(6):R834-R847. doi: 10.1152/ajpregu.00270.2017. Epub 2018 Jan 31. Am J Physiol Regul Integr Comp Physiol. 2018. PMID: 29384700 Free PMC article.
-
The Na/K-ATPase Signaling and SGLT2 Inhibitor-Mediated Cardiorenal Protection: A Crossed Road?J Membr Biol. 2021 Dec;254(5-6):513-529. doi: 10.1007/s00232-021-00192-z. Epub 2021 Jul 23. J Membr Biol. 2021. PMID: 34297135 Free PMC article. Review.
-
Targeting Na/K-ATPase Signaling: A New Approach to Control Oxidative Stress.Curr Pharm Des. 2018;24(3):359-364. doi: 10.2174/1381612824666180110101052. Curr Pharm Des. 2018. PMID: 29318961 Free PMC article. Review.
References
-
- Anderson DM, Williams DE, Tushinski R, Gimpel S, Eisenman J, Cannizzaro LA, Aronson M, Croce CM, Huebner K, Cosman D, Lyman SD. Alternate splicing of mRNAs encoding human mast cell growth factor and localization of the gene to chromosome 12q22-q24. Cell Growth Differ 2: 373–378, 1991 - PubMed
-
- Beltrami AP, Barlucchi L, Torella D, Baker M, Limana F, Chimenti S, Kasahara H, Rota M, Musso E, Urbanek K, Leri A, Kajstura J, Nadal-Ginard B, Anversa P. Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell 114: 763–776, 2003 - PubMed
-
- Bolli R, Chugh AR, D'Amario D, Loughran JH, Stoddard MF, Ikram S, Beache GM, Wagner SG, Leri A, Hosoda T, Sanada F, Elmore JB, Goichberg P, Cappetta D, Solankhi NK, Fahsah I, Rokosh DG, Slaughter MS, Kajstura J, Anversa P. Cardiac stem cells in patients with ischaemic cardiomyopathy (SCIPIO): initial results of a randomised phase 1 trial. Lancet 378: 1847–1857, 2011 - PMC - PubMed
-
- Diwan A, Dorn GW., 2nd Decompensation of cardiac hypertrophy: cellular mechanisms and novel therapeutic targets. Physiology (Bethesda) 22: 56–64, 2007 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

