Bronchial epithelial injury in the context of alloimmunity promotes lymphocytic bronchiolitis through hyaluronan expression
- PMID: 24748604
- PMCID: PMC4042191
- DOI: 10.1152/ajplung.00353.2013
Bronchial epithelial injury in the context of alloimmunity promotes lymphocytic bronchiolitis through hyaluronan expression
Abstract
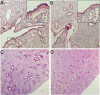
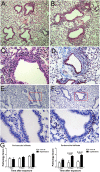
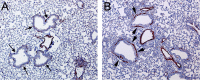
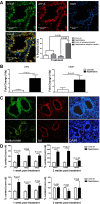
Epithelial injury is often detected in lung allografts, however, its relation to rejection pathogenesis is unknown. We hypothesized that sterile epithelial injury can lead to alloimmune activation in the lung. We performed adoptive transfer of mismatched splenocytes into recombinant activating gene 1 (Rag1)-deficient mice to induce an alloimmune status and then exposed these mice to naphthalene to induce sterile epithelial injury. We evaluated lungs for presence of alloimmune lung injury, endoplasmic reticulum (ER) stress, and hyaluronan expression, examined the effect of ER stress induction on hyaluronan expression and lymphocyte trapping by bronchial epithelia in vitro, and examined airways from patients with bronchiolitis obliterans syndrome and normal controls histologically. We found that Rag1-deficient mice that received mismatched splenocytes and naphthalene injection displayed bronchial epithelial ER stress, peribronchial hyaluronan expression, and lymphocytic bronchitis. Bronchial epithelial ER stress led to the expression of lymphocyte-trapping hyaluronan cables in vitro. Blockade of hyaluronan binding ameliorated naphthalene-induced lymphocytic bronchitis. ER stress was present histologically in >40% of bronchial epithelia of BOS patients and associated with subepithelial hyaluronan deposition. We conclude that sterile bronchial epithelial injury in the context of alloimmunity can lead to sustained ER stress and promote allograft rejection through hyaluronan expression.
Keywords: endoplasmic reticulum stress; lung rejection.
Copyright © 2014 the American Physiological Society.
Figures


Similar articles
-
Hyaluronan contributes to bronchiolitis obliterans syndrome and stimulates lung allograft rejection through activation of innate immunity.Am J Respir Crit Care Med. 2014 Mar 1;189(5):556-66. doi: 10.1164/rccm.201308-1481OC. Am J Respir Crit Care Med. 2014. PMID: 24471427 Free PMC article.
-
Ingraft chimerism in lung transplantation--a study in a porcine model of obliterative bronchiolitis.Respir Res. 2011 Apr 26;12(1):56. doi: 10.1186/1465-9921-12-56. Respir Res. 2011. PMID: 21521503 Free PMC article.
-
Epithelial tenascin predicts obliterative airway disease.J Heart Lung Transplant. 2008 Apr;27(4):400-7. doi: 10.1016/j.healun.2008.01.009. J Heart Lung Transplant. 2008. PMID: 18374876
-
Bronchiolitis obliterans: the Achilles heel of lung transplantation.Verh K Acad Geneeskd Belg. 2002;64(5):303-19; discussion 319-22. Verh K Acad Geneeskd Belg. 2002. PMID: 12647579 Review.
-
Bronchiolitis obliterans syndrome: alloimmune-dependent and -independent injury with aberrant tissue remodeling.Semin Thorac Cardiovasc Surg. 2008 Summer;20(2):173-82. doi: 10.1053/j.semtcvs.2008.05.002. Semin Thorac Cardiovasc Surg. 2008. PMID: 18707652 Review.
Cited by
-
Emerging concepts in smooth muscle contributions to airway structure and function: implications for health and disease.Am J Physiol Lung Cell Mol Physiol. 2016 Dec 1;311(6):L1113-L1140. doi: 10.1152/ajplung.00370.2016. Epub 2016 Oct 14. Am J Physiol Lung Cell Mol Physiol. 2016. PMID: 27742732 Free PMC article. Review.
-
Hyaluronan interactions with innate immunity in lung biology.Matrix Biol. 2019 May;78-79:84-99. doi: 10.1016/j.matbio.2018.01.027. Epub 2018 Feb 2. Matrix Biol. 2019. PMID: 29410190 Free PMC article. Review.
-
Sterile inflammation in thoracic transplantation.Cell Mol Life Sci. 2021 Jan;78(2):581-601. doi: 10.1007/s00018-020-03615-7. Epub 2020 Aug 17. Cell Mol Life Sci. 2021. PMID: 32803398 Free PMC article. Review.
-
The where, when, how, and why of hyaluronan binding by immune cells.Front Immunol. 2015 Apr 14;6:150. doi: 10.3389/fimmu.2015.00150. eCollection 2015. Front Immunol. 2015. PMID: 25926830 Free PMC article. Review.
-
High molecular weight hyaluronan ameliorates allergic inflammation and airway hyperresponsiveness in the mouse.Am J Physiol Lung Cell Mol Physiol. 2018 Nov 1;315(5):L787-L798. doi: 10.1152/ajplung.00009.2018. Epub 2018 Sep 6. Am J Physiol Lung Cell Mol Physiol. 2018. PMID: 30188746 Free PMC article.
References
-
- Borthwick LA, Parker SM, Brougham KA, Johnson GE, Gorowiec MR, Ward C, Lordan JL, Corris PA, Kirby JA, Fisher AJ. Epithelial to mesenchymal transition (EMT) and airway remodelling after human lung transplantation. Thorax 64: 770–777, 2009. - PubMed
-
- Buckpitt A, Chang AM, Weir A, Van Winkle L, Duan X, Philpot R, Plopper C. Relationship of cytochrome P450 activity to Clara cell cytotoxicity. IV. Metabolism of naphthalene and naphthalene oxide in microdissected airways from mice, rats, and hamsters. Mol Pharmacol 47: 74–81, 1995. - PubMed
-
- Chichester CH, Buckpitt AR, Chang A, Plopper CG. Metabolism and cytotoxicity of naphthalene and its metabolites in isolated murine Clara cells. Mol Pharmacol 45: 664–672, 1994. - PubMed
-
- Cho M, Chichester C, Morin D, Plopper C, Buckpitt A. Covalent interactions of reactive naphthalene metabolites with proteins. J Pharmacol Exp Ther 269: 881–889, 1994. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

