Gold nanoparticles enhanced electroporation for mammalian cell transfection
- PMID: 24749393
- PMCID: PMC4696415
- DOI: 10.1166/jbn.2014.1797
Gold nanoparticles enhanced electroporation for mammalian cell transfection
Abstract
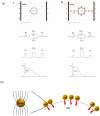
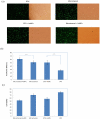
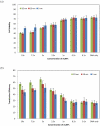
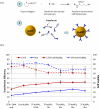
Electroporation figured prominently as an effective nonviral gene delivery approach for its balance on the transfection efficiency and cell viability, no restrictions of probe or cell type, and operation simplicity. The commercial electroporation systems have been widely adopted in the past two decades while still carry drawbacks associated with the high applied electric voltage, unsatisfied delivery efficiency, and/or low cell viability. By adding highly conductive gold nanoparticles (AuNPs) in electroporation solution, we demonstrated enhanced electroporation performance (i.e., better DNA delivery efficiency and higher cell viability) on mammalian cells from two different aspects: the free, naked AuNPs reduce the resistance of the electroporation solution so that the local pulse strength on cells was enhanced; targeting AuNPs (e.g., Tf-AuNPs) were brought to the cell membrane to work as virtual microelectrodes to porate cells with limited area from many different sites. The enhancement was confirmed with leukemia cells in both a commercial batch electroporation system and a home-made flow-through system using pWizGFP plasmid DNA probes. Such enhancement depends on the size, concentration, and the mixing ratio of free AuNPs/Tf-AuNPs. An equivalent mixture of free AuNPs and Tf-AuNPs exhibited the best enhancement with the transfection efficiency increased 2-3 folds at minimum sacrifice of cell viability. This new delivery concept, the combination of nanoparticles and electroporation technologies, may stimulate various in vitro and in vivo biomedical applications which rely on the efficient delivery of nucleic acids, anticancer drugs, or other therapeutic materials.
Figures




Similar articles
-
Gold nanoparticles electroporation enhanced polyplex delivery to mammalian cells.Electrophoresis. 2014 Jul;35(12-13):1837-45. doi: 10.1002/elps.201300617. Electrophoresis. 2014. PMID: 24777715 Free PMC article.
-
Gold nanoparticle-enhanced electroporation for leukemia cell transfection.Methods Mol Biol. 2014;1121:69-77. doi: 10.1007/978-1-4614-9632-8_6. Methods Mol Biol. 2014. PMID: 24510813
-
Fabrication Of Gold Nanoparticles In Absence Of Surfactant As In Vitro Carrier Of Plasmid DNA.Int J Nanomedicine. 2019 Oct 22;14:8399-8408. doi: 10.2147/IJN.S226498. eCollection 2019. Int J Nanomedicine. 2019. PMID: 31695373 Free PMC article.
-
Micro-/nano-electroporation for active gene delivery.Curr Pharm Des. 2015;21(42):6081-8. doi: 10.2174/1381612821666151027152121. Curr Pharm Des. 2015. PMID: 26503150 Review.
-
Biomedical Applications of DNA-Conjugated Gold Nanoparticles.Chembiochem. 2016 Jun 16;17(12):1052-62. doi: 10.1002/cbic.201600014. Epub 2016 Apr 8. Chembiochem. 2016. PMID: 26864481 Review.
Cited by
-
Electromagnetically Stimuli-Responsive Nanoparticles-Based Systems for Biomedical Applications: Recent Advances and Future Perspectives.Nanomaterials (Basel). 2021 Mar 26;11(4):848. doi: 10.3390/nano11040848. Nanomaterials (Basel). 2021. PMID: 33810343 Free PMC article. Review.
-
Localization of nanospheres in pheochromocytoma-like cells following exposure to high-frequency electromagnetic fields at 18 GHz.R Soc Open Sci. 2022 Jun 29;9(6):220520. doi: 10.1098/rsos.220520. eCollection 2022 Jun. R Soc Open Sci. 2022. PMID: 35774138 Free PMC article.
-
Gold nanoparticles electroporation enhanced polyplex delivery to mammalian cells.Electrophoresis. 2014 Jul;35(12-13):1837-45. doi: 10.1002/elps.201300617. Electrophoresis. 2014. PMID: 24777715 Free PMC article.
-
Lipoic Acid Gold Nanoparticles Functionalized with Organic Compounds as Bioactive Materials.Nanomaterials (Basel). 2017 Feb 16;7(2):43. doi: 10.3390/nano7020043. Nanomaterials (Basel). 2017. PMID: 28336877 Free PMC article.
-
Immunocyte membrane-derived biomimetic nano-drug delivery system: a pioneering platform for tumour immunotherapy.Acta Pharmacol Sin. 2024 Dec;45(12):2455-2473. doi: 10.1038/s41401-024-01355-z. Epub 2024 Jul 31. Acta Pharmacol Sin. 2024. PMID: 39085407 Review.
References
-
- Luo D, Saltzman WM. Synthetic DNA delivery systems. Nat. Biotechnol. 2000;18:33–37. - PubMed
-
- Templeton NS. Editor, Gene and cell therapy: therapeutic mechanism and strategies. 2nd Marcel Dekker; New York: 2004.
-
- Hamer DH, Leder P. Splicing and the formation of stable RNA. Cell. 1979;18:1299–1302. - PubMed
-
- Mulligan RC, Howard BH, Berg P. Synthesis of rabbit beta-globin in cultured monkey kidney cells following infection with a SV40 beta-globin recombinant genome. Nature. 1979;277:108–114. - PubMed
-
- Verma IM, Somia N. Gene therapy -- promises, problems and prospects. Nature. 1997;389:239–242. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
