Crystal structure of the Campylobacter jejuni CmeC outer membrane channel
- PMID: 24753291
- PMCID: PMC4088979
- DOI: 10.1002/pro.2478
Crystal structure of the Campylobacter jejuni CmeC outer membrane channel
Abstract
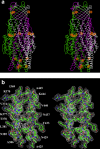
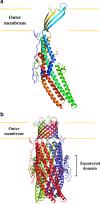
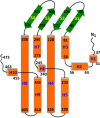
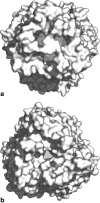
As one of the world's most prevalent enteric pathogens, Campylobacter jejuni is a major causative agent of human enterocolitis and is responsible for more than 400 million cases of diarrhea each year. The impact of this pathogen on children is of particular significance. Campylobacter has developed resistance to many antimicrobial agents via multidrug efflux machinery. The CmeABC tripartite multidrug efflux pump, belonging to the resistance-nodulation-cell division (RND) superfamily, plays a major role in drug resistant phenotypes of C. jejuni. This efflux complex spans the entire cell envelop of C. jejuni and mediates resistance to various antibiotics and toxic compounds. We here report the crystal structure of C. jejuni CmeC, the outer membrane component of the CmeABC tripartite multidrug efflux system. The structure reveals a possible mechanism for substrate export.
Keywords: Campylobacter jejuni; efflux channel; membrane protein; multidrug resistance; resistance-nodulation-cell division.
© 2014 The Protein Society.
Figures


References
-
- Ruiz-Palacios GM. The health burden of Campylobacter infection and the impact of antimicrobial resistance: Playing chicken. Clin Infect Dis. 2007;44:701–703. - PubMed
-
- van Doorn PA, Ruts L, Jacobs BC. Clinical features, pathogenesis, and treatment of Guillain-Barre syndrome. Lancet Neurol. 2008;7:939–950. - PubMed
-
- Gibreel A, Taylor DE. Macrolide resistance in Campylobacter jejuni and Campylobacter coli. J Antimicrob Chemother. 2006;58:243–255. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

