Comparative study of the shell development of hard- and soft-shelled turtles
- PMID: 24754673
- PMCID: PMC4089346
- DOI: 10.1111/joa.12189
Comparative study of the shell development of hard- and soft-shelled turtles
Abstract
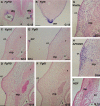
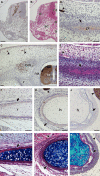
The turtle shell provides a fascinating model for the investigation of the evolutionary modifications of developmental mechanisms. Different conclusions have been put forth for its development, and it is suggested that one of the causes of the disagreement could be the differences in the species of the turtles used - the differences between hard-shelled turtles and soft-shelled turtles. To elucidate the cause of the difference, we compared the turtle shell development in the two groups of turtle. In the dorsal shell development, these two turtle groups shared the gene expression profile that is required for formation, and shared similar spatial organization of the anatomical elements during development. Thus, both turtles formed the dorsal shell through a folding of the lateral body wall, and the Wnt signaling pathway appears to have been involved in the development. The ventral portion of the shell, on the other hand, contains massive dermal bones. Although expression of HNK-1 epitope has suggested that the trunk neural crest contributed to the dermal bones in the hard-shelled turtles, it was not expressed in the initial anlage of the skeletons in either of the types of turtle. Hence, no evidence was found that would support a neural crest origin.
Keywords: carapace; marker genes; neural crest; plastron; turtles.
© 2014 Anatomical Society.
Figures





Similar articles
-
Comparative study of collagen distribution in the dermis of the embryonic carapace of soft- and hard-shelled cryptodiran turtles.J Morphol. 2021 Apr;282(4):543-552. doi: 10.1002/jmor.21327. Epub 2021 Feb 8. J Morphol. 2021. PMID: 33491791
-
Evidence for the neural crest origin of turtle plastron bones.Genesis. 2001 Nov;31(3):111-7. doi: 10.1002/gene.10012. Genesis. 2001. PMID: 11747201
-
The origin and loss of periodic patterning in the turtle shell.Development. 2014 Aug;141(15):3033-9. doi: 10.1242/dev.109041. Development. 2014. PMID: 25053434
-
The evolutionary origin of the turtle shell and its dependence on the axial arrest of the embryonic rib cage.J Exp Zool B Mol Dev Evol. 2015 May;324(3):194-207. doi: 10.1002/jez.b.22579. Epub 2014 Jun 5. J Exp Zool B Mol Dev Evol. 2015. PMID: 24898540 Review.
-
Body plan of turtles: an anatomical, developmental and evolutionary perspective.Anat Sci Int. 2012 Mar;87(1):1-13. doi: 10.1007/s12565-011-0121-y. Epub 2011 Dec 1. Anat Sci Int. 2012. PMID: 22131042 Review.
Cited by
-
Gene Regulation during Carapacial Ridge Development of Mauremys reevesii: The Development of Carapacial Ridge, Ribs and Scutes.Genes (Basel). 2022 Sep 19;13(9):1676. doi: 10.3390/genes13091676. Genes (Basel). 2022. PMID: 36140843 Free PMC article.
-
Delayed trait development and the convergent evolution of shell kinesis in turtles.Proc Biol Sci. 2018 Oct 3;285(1888):20181585. doi: 10.1098/rspb.2018.1585. Proc Biol Sci. 2018. PMID: 30282655 Free PMC article.
-
Cranial neural crest cell contribution to craniofacial formation, pathology, and future directions in tissue engineering.Birth Defects Res C Embryo Today. 2014 Sep;102(3):324-32. doi: 10.1002/bdrc.21075. Epub 2014 Sep 16. Birth Defects Res C Embryo Today. 2014. PMID: 25227212 Free PMC article. Review.
-
Skeletal remodelling suggests the turtle's shell is not an evolutionary straitjacket.Biol Lett. 2015 Apr;11(4):20150022. doi: 10.1098/rsbl.2015.0022. Biol Lett. 2015. PMID: 25878046 Free PMC article.
-
Global Analysis of Transcriptome and Translatome Revealed That Coordinated WNT and FGF Regulate the Carapacial Ridge Development of Chinese Soft-Shell Turtle.Int J Mol Sci. 2021 Nov 18;22(22):12441. doi: 10.3390/ijms222212441. Int J Mol Sci. 2021. PMID: 34830331 Free PMC article.
References
-
- Abo T, Balch CM. A differentiation antigen of human NK and K cells identified by a monoclonal antibody (HNK-1) J Immunol. 1981;127:1024–1029. - PubMed
-
- Abzhanov A, Tzahor E, Lassar AB, et al. Dissimilar regulation of cell differentiation in mesencephalic (cranial) and sacral (trunk) neural crest cells in vitro. Development. 2003;130:4567–4579. - PubMed
-
- Bronner-Fraser M. Analysis of the early stages of trunk neural crest migration in avian embryos using monoclonal antibody HNK-1. Dev Biol. 1986;115:44–55. - PubMed
-
- Burke AC. Development of the turtle carapace: implications for the evolution of a novel bauplan. J Morphol. 1989;199:363–378. - PubMed
-
- Burke AC. The development and evolution of the turtle body plan. Inferring intrinsic aspects of the evolutionary process from experimental embryology. Am Zool. 1991;31:616–627.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

