Inhibition of Rab5 Activation During Insulin Receptor-Mediated Endocytosis
- PMID: 24765621
- PMCID: PMC3995085
Inhibition of Rab5 Activation During Insulin Receptor-Mediated Endocytosis
Abstract
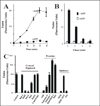
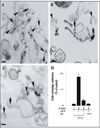
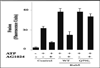
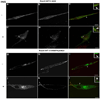
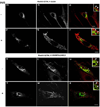
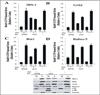
Activation of receptor tyrosine kinases is a key feature in receptor signaling and membrane trafficking processes. In this study, we found that the insulin receptor tyrosine kinase activity is required for fusion between early endosomes. AG1024, a receptor tyrosine kinase inhibitor, blocked the in vitro endosome fusion in a concentration-dependent manner. We observed that Rab5: wild type partially rescued the fusion reaction, whereas Rab5: Q79L mutant fully rescued it. We also observed that treatment of cells with insulin receptor kinase inhibitor HNMPA-(AM)3 blocked the formation of Rab5-positive endosomes as well as the activation of Rab5 upon addition of insulin in intact cells. HNMPA-(AM)3 inhibitor also affected the endosomal co-localization of Rab5 and insulin receptor. However, the formation of Rab5: Q79L mutant-positive endosomes were not affected by the HNMPA-(AM)3 inhibitor. In addition, HNMPA-(AM)3 inhibitor affected the association of Rin1 to membrane upon insulin stimulation. Furthermore, Rin1 did not fully support endosome fusion in the presence of the AG1024 inhibitor. These results constitute the first evidence that, at least in part, the enzymatic activity of insulin receptor is required for the fusion events via the activation of Rab5.
Keywords: Endosome fusion; Kinase Inhibitors; Receptor tyrosine kinase; small GTPases.
Figures




Similar articles
-
Effect of EGF-receptor tyrosine kinase inhibitor on Rab5 function during endocytosis.Arch Biochem Biophys. 2012 Sep 1;525(1):16-24. doi: 10.1016/j.abb.2012.05.023. Epub 2012 Jun 5. Arch Biochem Biophys. 2012. PMID: 22683472 Free PMC article.
-
Evidence for phosphatidylinositol 3-kinase as a regulator of endocytosis via activation of Rab5.Proc Natl Acad Sci U S A. 1995 Oct 24;92(22):10207-11. doi: 10.1073/pnas.92.22.10207. Proc Natl Acad Sci U S A. 1995. PMID: 7479754 Free PMC article.
-
Evidence for a symmetrical requirement for Rab5-GTP in in vitro endosome-endosome fusion.J Biol Chem. 1998 Oct 2;273(40):25850-5. doi: 10.1074/jbc.273.40.25850. J Biol Chem. 1998. PMID: 9748259
-
Vps34 and the Armus/TBC-2 Rab GAPs: Putting the brakes on the endosomal Rab5 and Rab7 GTPases.Cell Logist. 2017 Dec 19;7(4):e1403530. doi: 10.1080/21592799.2017.1403530. eCollection 2017. Cell Logist. 2017. PMID: 29296513 Free PMC article. Review.
-
The Emerging Role of Rab5 in Membrane Receptor Trafficking and Signaling Pathways.Biochem Res Int. 2020 Feb 11;2020:4186308. doi: 10.1155/2020/4186308. eCollection 2020. Biochem Res Int. 2020. PMID: 32104603 Free PMC article. Review.
Cited by
-
Stress Signals, Mediated by Membranous Glucocorticoid Receptor, Activate PLC/PKC/GSK-3β/β-catenin Pathway to Inhibit Wound Closure.J Invest Dermatol. 2017 May;137(5):1144-1154. doi: 10.1016/j.jid.2016.11.036. Epub 2016 Dec 23. J Invest Dermatol. 2017. PMID: 28017831 Free PMC article.
-
Protein kinase- and lipase inhibitors of inositide metabolism deplete IP7 indirectly in pancreatic β-cells: Off-target effects on cellular bioenergetics and direct effects on IP6K activity.Cell Signal. 2018 Jan;42:127-133. doi: 10.1016/j.cellsig.2017.10.008. Epub 2017 Oct 16. Cell Signal. 2018. PMID: 29042286 Free PMC article.
-
Glucocorticoid-mediated induction of caveolin-1 disrupts cytoskeletal organization, inhibits cell migration and re-epithelialization of non-healing wounds.Commun Biol. 2021 Jun 18;4(1):757. doi: 10.1038/s42003-021-02298-5. Commun Biol. 2021. PMID: 34145387 Free PMC article.
References
-
- Barbieri MA, Hoffenberg S, Roberts R, Mukhopadhyay A, Pomrehn A, Dickey BF, Stahl PD. Evidence for a symmetrical requirement for Rab5-GTP in in vitro endosome-endosome fusion. Journal of Biological Chemistry. 1998;273:25850–25855. - PubMed
-
- Barbieri MA, Li G, Colombo MI, Stahl PD. Rab5, an early acting endosomal GTPase, supports in vitro endosome fusion without GTP hydrolysis. Journal of Biological Chemistry. 1994;269:18720–18722. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
