ConSole: using modularity of contact maps to locate solenoid domains in protein structures
- PMID: 24766872
- PMCID: PMC4021314
- DOI: 10.1186/1471-2105-15-119
ConSole: using modularity of contact maps to locate solenoid domains in protein structures
Abstract
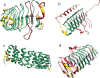
Background: Periodic proteins, characterized by the presence of multiple repeats of short motifs, form an interesting and seldom-studied group. Due to often extreme divergence in sequence, detection and analysis of such motifs is performed more reliably on the structural level. Yet, few algorithms have been developed for the detection and analysis of structures of periodic proteins.
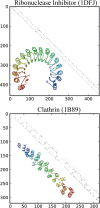
Results: ConSole recognizes modularity in protein contact maps, allowing for precise identification of repeats in solenoid protein structures, an important subgroup of periodic proteins. Tests on benchmarks show that ConSole has higher recognition accuracy as compared to Raphael, the only other publicly available solenoid structure detection tool. As a next step of ConSole analysis, we show how detection of solenoid repeats in structures can be used to improve sequence recognition of these motifs and to detect subtle irregularities of repeat lengths in three solenoid protein families.
Conclusions: The ConSole algorithm provides a fast and accurate tool to recognize solenoid protein structures as a whole and to identify individual solenoid repeat units from a structure. ConSole is available as a web-based, interactive server and is available for download at http://console.sanfordburnham.org.
Figures



Similar articles
-
RAPHAEL: recognition, periodicity and insertion assignment of solenoid protein structures.Bioinformatics. 2012 Dec 15;28(24):3257-64. doi: 10.1093/bioinformatics/bts550. Epub 2012 Sep 8. Bioinformatics. 2012. PMID: 22962341
-
Revealing aperiodic aspects of solenoid proteins from sequence information.Bioinformatics. 2016 Sep 15;32(18):2776-82. doi: 10.1093/bioinformatics/btw319. Epub 2016 Jun 9. Bioinformatics. 2016. PMID: 27334472 Free PMC article.
-
REPETITA: detection and discrimination of the periodicity of protein solenoid repeats by discrete Fourier transform.Bioinformatics. 2009 Jun 15;25(12):i289-95. doi: 10.1093/bioinformatics/btp232. Bioinformatics. 2009. PMID: 19478001 Free PMC article.
-
When protein folding is simplified to protein coiling: the continuum of solenoid protein structures.Trends Biochem Sci. 2000 Oct;25(10):509-15. doi: 10.1016/s0968-0004(00)01667-4. Trends Biochem Sci. 2000. PMID: 11050437 Review.
-
Protein repeats: structures, functions, and evolution.J Struct Biol. 2001 May-Jun;134(2-3):117-31. doi: 10.1006/jsbi.2001.4392. J Struct Biol. 2001. PMID: 11551174 Review.
Cited by
-
NAPS: Network Analysis of Protein Structures.Nucleic Acids Res. 2016 Jul 8;44(W1):W375-82. doi: 10.1093/nar/gkw383. Epub 2016 May 5. Nucleic Acids Res. 2016. PMID: 27151201 Free PMC article.
-
Quantitative analysis of visual codewords of a protein distance matrix.PLoS One. 2022 Feb 4;17(2):e0263566. doi: 10.1371/journal.pone.0263566. eCollection 2022. PLoS One. 2022. PMID: 35120181 Free PMC article.
-
RepeatsDB 2.0: improved annotation, classification, search and visualization of repeat protein structures.Nucleic Acids Res. 2017 Jan 4;45(D1):D308-D312. doi: 10.1093/nar/gkw1136. Epub 2016 Nov 29. Nucleic Acids Res. 2017. PMID: 27899671 Free PMC article.
-
Tandem Repeats in Proteins: Prediction Algorithms and Biological Role.Front Bioeng Biotechnol. 2015 Sep 24;3:143. doi: 10.3389/fbioe.2015.00143. eCollection 2015. Front Bioeng Biotechnol. 2015. PMID: 26442257 Free PMC article. Review.
-
Analyzing the symmetrical arrangement of structural repeats in proteins with CE-Symm.PLoS Comput Biol. 2019 Apr 22;15(4):e1006842. doi: 10.1371/journal.pcbi.1006842. eCollection 2019 Apr. PLoS Comput Biol. 2019. PMID: 31009453 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

