Lhx1 controls terminal differentiation and circadian function of the suprachiasmatic nucleus
- PMID: 24767996
- PMCID: PMC4254772
- DOI: 10.1016/j.celrep.2014.03.060
Lhx1 controls terminal differentiation and circadian function of the suprachiasmatic nucleus
Abstract
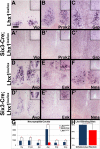
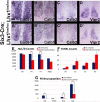
Vertebrate circadian rhythms are organized by the hypothalamic suprachiasmatic nucleus (SCN). Despite its physiological importance, SCN development is poorly understood. Here, we show that Lim homeodomain transcription factor 1 (Lhx1) is essential for terminal differentiation and function of the SCN. Deletion of Lhx1 in the developing SCN results in loss of SCN-enriched neuropeptides involved in synchronization and coupling to downstream oscillators, among other aspects of circadian function. Intact, albeit damped, clock gene expression rhythms persist in Lhx1-deficient SCN; however, circadian activity rhythms are highly disorganized and susceptible to surprising changes in period, phase, and consolidation following neuropeptide infusion. Our results identify a factor required for SCN terminal differentiation. In addition, our in vivo study of combinatorial SCN neuropeptide disruption uncovered synergies among SCN-enriched neuropeptides in regulating normal circadian function. These animals provide a platform for studying the central oscillator's role in physiology and cognition.
Copyright © 2014 The Authors. Published by Elsevier Inc. All rights reserved.
Figures



References
-
- Abrahamson EE, Moore RY. Suprachiasmatic nucleus in the mouse: retinal innervation, intrinsic organization and efferent projections. Brain Res. 2001;916:172–191. - PubMed
-
- Albers HE, Gillespie CF, Babagbemi TO, Huhman KL. Analysis of the phase shifting effects of gastrin releasing peptide when microinjected into the suprachiasmatic region. Neurosci. Lett. 1995;191:63–66. - PubMed
-
- Albrecht U. Timing to perfection: the biology of central and peripheral circadian clocks. Neuron. 2012;74:246–260. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

