An increase in tolerogenic dendritic cell and natural regulatory T cell numbers during experimental autoimmune encephalomyelitis in Rras-/- mice results in attenuated disease
- PMID: 24771856
- PMCID: PMC4041102
- DOI: 10.4049/jimmunol.1302254
An increase in tolerogenic dendritic cell and natural regulatory T cell numbers during experimental autoimmune encephalomyelitis in Rras-/- mice results in attenuated disease
Abstract
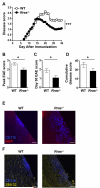
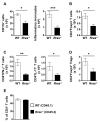
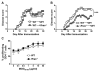
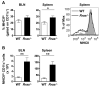
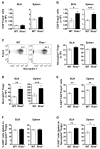
R-Ras is a member of the Ras superfamily of small GTPases, which are regulators of various cellular processes, including adhesion, survival, proliferation, trafficking, and cytokine production. R-Ras is expressed by immune cells and has been shown to modulate dendritic cell (DC) function in vitro and has been associated with liver autoimmunity. We used Rras-deficient mice to study the mechanism whereby R-Ras contributes to autoimmunity using experimental autoimmune encephalomyelitis (EAE), a mouse model of the CNS autoimmune disease multiple sclerosis. We found that a lack of R-Ras in peripheral immune cells resulted in attenuated EAE disease. Further investigation revealed that, during EAE, absence of R-Ras promoted the formation of MHC II(low) DC concomitant with a significant increase in proliferation of natural regulatory T cells, resulting in an increase in their cell numbers in the periphery. Our study suggests a novel role for R-Ras in promoting autoimmunity through negative regulation of natural regulatory T cell numbers by inhibiting the development of MHCII(low) DC with tolerogenic potential.
Copyright © 2014 by The American Association of Immunologists, Inc.
Figures
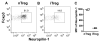
Similar articles
-
Neuroantigen-specific autoregulatory CD8+ T cells inhibit autoimmune demyelination through modulation of dendritic cell function.PLoS One. 2014 Aug 21;9(8):e105763. doi: 10.1371/journal.pone.0105763. eCollection 2014. PLoS One. 2014. PMID: 25144738 Free PMC article.
-
Early life exposure to lipopolysaccharide suppresses experimental autoimmune encephalomyelitis by promoting tolerogenic dendritic cells and regulatory T cells.J Immunol. 2009 Jul 1;183(1):298-309. doi: 10.4049/jimmunol.0803576. J Immunol. 2009. PMID: 19542441
-
Galectin-1 is essential for the induction of MOG35-55 -based intravenous tolerance in experimental autoimmune encephalomyelitis.Eur J Immunol. 2016 Jul;46(7):1783-96. doi: 10.1002/eji.201546212. Epub 2016 May 25. Eur J Immunol. 2016. PMID: 27151444 Free PMC article.
-
Emerging concepts in autoimmune encephalomyelitis beyond the CD4/T(H)1 paradigm.Ann Anat. 2010 Aug 20;192(4):179-93. doi: 10.1016/j.aanat.2010.06.006. Epub 2010 Jul 15. Ann Anat. 2010. PMID: 20692821 Review.
-
The role of regulatory T cells in multiple sclerosis.Nat Clin Pract Neurol. 2008 Jul;4(7):384-98. doi: 10.1038/ncpneuro0832. Epub 2008 Jun 24. Nat Clin Pract Neurol. 2008. PMID: 18578001 Review.
Cited by
-
Interrelatedness between dysbiosis in the gut microbiota due to immunodeficiency and disease penetrance of colitis.Immunology. 2015 Nov;146(3):359-68. doi: 10.1111/imm.12511. Epub 2015 Sep 7. Immunology. 2015. PMID: 26211540 Free PMC article. Review.
-
R-Ras deficiency does not affect papain-induced IgE production in mice.Immun Inflamm Dis. 2017 Sep;5(3):280-288. doi: 10.1002/iid3.168. Epub 2017 May 11. Immun Inflamm Dis. 2017. PMID: 28497586 Free PMC article.
-
The Role of R-Ras Proteins in Normal and Pathologic Migration and Morphologic Change.Am J Pathol. 2021 Sep;191(9):1499-1510. doi: 10.1016/j.ajpath.2021.05.008. Epub 2021 Jun 7. Am J Pathol. 2021. PMID: 34111428 Free PMC article. Review.
-
Small GTPase RAS in multiple sclerosis - exploring the role of RAS GTPase in the etiology of multiple sclerosis.Small GTPases. 2020 Sep;11(5):312-319. doi: 10.1080/21541248.2018.1502591. Epub 2018 Sep 18. Small GTPases. 2020. PMID: 30043672 Free PMC article. Review.
-
Inhibition of myeloperoxidase at the peak of experimental autoimmune encephalomyelitis restores blood-brain barrier integrity and ameliorates disease severity.J Neurochem. 2016 Feb;136(4):826-836. doi: 10.1111/jnc.13426. Epub 2015 Nov 30. J Neurochem. 2016. PMID: 26560636 Free PMC article.
References
-
- Zhang Z, Vuori K, Wang H, Reed JC, Ruoslahti E. Integrin activation by R-ras. Cell. 1996;85:61–69. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

