Collagen-binding protein, Aegyptin, regulates probing time and blood feeding success in the dengue vector mosquito, Aedes aegypti
- PMID: 24778255
- PMCID: PMC4024861
- DOI: 10.1073/pnas.1404179111
Collagen-binding protein, Aegyptin, regulates probing time and blood feeding success in the dengue vector mosquito, Aedes aegypti
Abstract
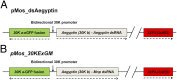
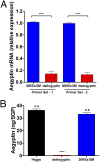
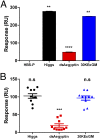
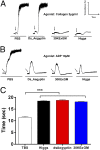
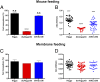
Mosquito salivary glands have important roles in blood feeding and pathogen transmission. However, the biological relevance of many salivary components has yet to be determined. Aegyptin, a secreted salivary protein from Aedes aegypti, binds collagen and inhibits platelet aggregation and adhesion. We used a transgenic approach to study the relevance of Aegyptin in mosquito blood feeding. Aedes aegypti manipulated genetically to express gene-specific inverted-repeat RNA sequences exhibited significant reductions in Aegyptin mRNA accumulation (85-87%) and protein levels (>80-fold) in female mosquito salivary glands. Transgenic mosquitoes had longer probing times (78-300 s, P < 0.0001) when feeding on mice compared with controls (15-56 s), feeding success was reduced, and those feeding took smaller blood meals. However, no differences in feeding success or blood meal size were found in membrane feeding experiments using defibrinated human blood. Salivary gland extracts from transgenic mosquitoes failed to inhibit collagen-induced platelet aggregation in vitro. Reductions of Aegyptin did not affect salivary ADP-induced platelet aggregation inhibition or disturb anticlotting activities. Our results demonstrate the relevance of Aegyptin for A. aegypti blood feeding, providing further support for the hypothesis that platelet aggregation inhibition is a vital salivary function in blood feeding arthropods. It has been suggested that the multiple mosquito salivary components mediating platelet aggregation (i.e., Aegyptin, apyrase, D7) represent functional redundancy. Our findings do not support this hypothesis; instead, they indicate that multiple salivary components work synergistically and are necessary to achieve maximum blood feeding efficiency.
Keywords: RNAi; evolution; hematophagy; saliva; transgenesis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Biochemical characterization of AeD7L2 and its physiological relevance in blood feeding in the dengue mosquito vector, Aedes aegypti.FEBS J. 2021 Mar;288(6):2014-2029. doi: 10.1111/febs.15524. Epub 2020 Sep 5. FEBS J. 2021. PMID: 32799410 Free PMC article.
-
Aegyptin, a novel mosquito salivary gland protein, specifically binds to collagen and prevents its interaction with platelet glycoprotein VI, integrin alpha2beta1, and von Willebrand factor.J Biol Chem. 2007 Sep 14;282(37):26928-26938. doi: 10.1074/jbc.M705669200. Epub 2007 Jul 24. J Biol Chem. 2007. PMID: 17650501 Free PMC article.
-
Aegyptin displays high-affinity for the von Willebrand factor binding site (RGQOGVMGF) in collagen and inhibits carotid thrombus formation in vivo.FEBS J. 2010 Jan;277(2):413-27. doi: 10.1111/j.1742-4658.2009.07494.x. Epub 2009 Dec 15. FEBS J. 2010. PMID: 20015075 Free PMC article.
-
Platelet aggregation inhibitors from hematophagous animals.Toxicon. 2010 Dec 15;56(7):1130-44. doi: 10.1016/j.toxicon.2009.12.003. Epub 2009 Dec 24. Toxicon. 2010. PMID: 20035779 Free PMC article. Review.
-
Unraveling the Molecular Mechanisms of Mosquito Salivary Proteins: New Frontiers in Disease Transmission and Control.Biomolecules. 2025 Jan 8;15(1):82. doi: 10.3390/biom15010082. Biomolecules. 2025. PMID: 39858476 Free PMC article. Review.
Cited by
-
Aedes aegypti post-emergence transcriptome: Unveiling the molecular basis for the hematophagic and gonotrophic capacitation.PLoS Negl Trop Dis. 2021 Jan 6;15(1):e0008915. doi: 10.1371/journal.pntd.0008915. eCollection 2021 Jan. PLoS Negl Trop Dis. 2021. PMID: 33406161 Free PMC article.
-
Vertebrate Responses against Arthropod Salivary Proteins and Their Therapeutic Potential.Vaccines (Basel). 2021 Apr 5;9(4):347. doi: 10.3390/vaccines9040347. Vaccines (Basel). 2021. PMID: 33916367 Free PMC article. Review.
-
Evaluation of Aedes aegypti Feeding Status.Cold Spring Harb Protoc. 2023 Jun 1;2023(6):pdb.prot108023. doi: 10.1101/pdb.prot108023. Cold Spring Harb Protoc. 2023. PMID: 36690389 Free PMC article.
-
Guianensin, a Simulium guianense salivary protein, has broad anti-hemostatic and anti-inflammatory properties.Front Immunol. 2023 Jul 3;14:1163367. doi: 10.3389/fimmu.2023.1163367. eCollection 2023. Front Immunol. 2023. PMID: 37469515 Free PMC article.
-
Mosquito salivary allergen Aed a 3: cloning, comprehensive molecular analysis, and clinical evaluation.Allergy. 2016 May;71(5):621-8. doi: 10.1111/all.12812. Epub 2016 Mar 6. Allergy. 2016. PMID: 26608594 Free PMC article.
References
-
- Ribeiro JMC, Rossignol PA, Spielman A. Aedes aegypti: Model for blood finding strategy and prediction of parasite manipulation. Exp Parasitol. 1985;60(1):118–132. - PubMed
-
- Kauskot A, Hoylaerts MF. Platelet receptors. Handb Exp Pharmacol. 2012;(210):23–57. - PubMed
-
- Hoffman R. Hematology: Basic Principles and Practice. 3rd Ed. New York: Churchill-Livingstone; 2000. p. 1783.
-
- Ribeiro JMC, Arca B. From sialomes to the sialoverse: An insight into the salivary potion of blood feeding insects. Adv Insect Physiol. 2009;37(2):59–118.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

