Curcumin attenuates angiogenesis in liver fibrosis and inhibits angiogenic properties of hepatic stellate cells
- PMID: 24779927
- PMCID: PMC4124023
- DOI: 10.1111/jcmm.12286
Curcumin attenuates angiogenesis in liver fibrosis and inhibits angiogenic properties of hepatic stellate cells
Abstract
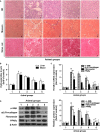
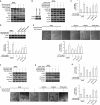
Hepatic fibrosis is concomitant with sinusoidal pathological angiogenesis, which has been highlighted as novel therapeutic targets for the treatment of chronic liver disease. Our prior studies have demonstrated that curcumin has potent antifibrotic activity, but the mechanisms remain to be elucidated. The current work demonstrated that curcumin ameliorated fibrotic injury and sinusoidal angiogenesis in rat liver with fibrosis caused by carbon tetrachloride. Curcumin reduced the expression of a number of angiogenic markers in fibrotic liver. Experiments in vitro showed that the viability and vascularization of rat liver sinusoidal endothelial cells and rat aortic ring angiogenesis were not impaired by curcumin. These results indicated that hepatic stellate cells (HSCs) that are characterized as liver-specific pericytes could be potential target cells for curcumin. Further investigations showed that curcumin inhibited VEGF expression in HSCs associated with disrupting platelet-derived growth factor-β receptor (PDGF-βR)/ERK and mTOR pathways. HSC motility and vascularization were also suppressed by curcumin associated with blocking PDGF-βR/focal adhesion kinase/RhoA cascade. Gain- or loss-of-function analyses revealed that activation of peroxisome proliferator-activated receptor-γ (PPAR-γ) was required for curcumin to inhibit angiogenic properties of HSCs. We concluded that curcumin attenuated sinusoidal angiogenesis in liver fibrosis possibly by targeting HSCs via a PPAR-γ activation-dependent mechanism. PPAR-γ could be a target molecule for reducing pathological angiogenesis during liver fibrosis.
Keywords: VEGF; angiogenesis; curcumin; hepatic stellate cell; liver fibrosis; peroxisome proliferator-activated receptor-γ.
© 2014 The Authors. Journal of Cellular and Molecular Medicine published by John Wiley & Sons Ltd and Foundation for Cellular and Molecular Medicine.
Figures





Similar articles
-
Peroxisome proliferator-activated receptor-γ interrupts angiogenic signal transduction by transrepression of platelet-derived growth factor-β receptor in hepatic stellate cells.J Cell Sci. 2014 Jan 15;127(Pt 2):305-14. doi: 10.1242/jcs.128306. Epub 2013 Nov 20. J Cell Sci. 2014. PMID: 24259663
-
Curcumin up-regulates phosphatase and tensin homologue deleted on chromosome 10 through microRNA-mediated control of DNA methylation--a novel mechanism suppressing liver fibrosis.FEBS J. 2014 Jan;281(1):88-103. doi: 10.1111/febs.12574. Epub 2013 Nov 5. FEBS J. 2014. PMID: 24138392
-
Canonical hedgehog signalling regulates hepatic stellate cell-mediated angiogenesis in liver fibrosis.Br J Pharmacol. 2017 Mar;174(5):409-423. doi: 10.1111/bph.13701. Epub 2017 Jan 31. Br J Pharmacol. 2017. PMID: 28052321 Free PMC article.
-
Peroxisome proliferator-activated receptor-γ as a therapeutic target for hepatic fibrosis: from bench to bedside.Cell Mol Life Sci. 2013 Jan;70(2):259-76. doi: 10.1007/s00018-012-1046-x. Epub 2012 Jun 15. Cell Mol Life Sci. 2013. PMID: 22699820 Free PMC article. Review.
-
Targeting Hepatic Stellate Cell Death to Reverse Hepatic Fibrosis.Curr Drug Targets. 2023;24(7):568-583. doi: 10.2174/1389450124666230330135834. Curr Drug Targets. 2023. PMID: 36999702 Review.
Cited by
-
Activation of PPARγ/P53 signaling is required for curcumin to induce hepatic stellate cell senescence.Cell Death Dis. 2016 Apr 14;7(4):e2189. doi: 10.1038/cddis.2016.92. Cell Death Dis. 2016. PMID: 27077805 Free PMC article.
-
Bisdemethoxycurcumin Induces apoptosis in activated hepatic stellate cells via cannabinoid receptor 2.Molecules. 2015 Jan 14;20(1):1277-92. doi: 10.3390/molecules20011277. Molecules. 2015. PMID: 25594342 Free PMC article.
-
Molecular Mechanisms and Potential New Therapeutic Drugs for Liver Fibrosis.Front Pharmacol. 2022 Feb 11;13:787748. doi: 10.3389/fphar.2022.787748. eCollection 2022. Front Pharmacol. 2022. PMID: 35222022 Free PMC article. Review.
-
Curcumin Interferes with TGF- β 1-Induced Fibrosis in NRK-49F Cells by Reversing ADAMTS18 Gene Methylation.Chin J Integr Med. 2024 Jul;30(7):600-607. doi: 10.1007/s11655-023-3564-9. Epub 2023 Sep 27. Chin J Integr Med. 2024. PMID: 37812341
-
Curcumin and chronic kidney disease (CKD): major mode of action through stimulating endogenous intestinal alkaline phosphatase.Molecules. 2014 Dec 2;19(12):20139-56. doi: 10.3390/molecules191220139. Molecules. 2014. PMID: 25474287 Free PMC article. Review.
References
-
- Valfre di Bonzo L, Novo E, Cannito S, et al. Angiogenesis and liver fibrogenesis. Histol Histopathol. 2009;24:1323–41. - PubMed
-
- Hernandez-Gea V, Friedman SL. Pathogenesis of liver fibrosis. Annu Rev Pathol. 2011;6:425–56. - PubMed
-
- Lee JS, Semela D, Iredale J, et al. Sinusoidal remodeling and angiogenesis: a new function for the liver-specific pericyte? Hepatology. 2007;45:817–25. - PubMed
-
- Thabut D, Shah V. Intrahepatic angiogenesis and sinusoidal remodeling in chronic liver disease: new targets for the treatment of portal hypertension? J Hepatol. 2010;53:976–80. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

