Early discharge using single cardiac troponin and copeptin testing in patients with suspected acute coronary syndrome (ACS): a randomized, controlled clinical process study
- PMID: 24786301
- PMCID: PMC4320319
- DOI: 10.1093/eurheartj/ehu178
Early discharge using single cardiac troponin and copeptin testing in patients with suspected acute coronary syndrome (ACS): a randomized, controlled clinical process study
Abstract
Aims: This randomized controlled trial (RCT) evaluated whether a process with single combined testing of copeptin and troponin at admission in patients with low-to-intermediate risk and suspected acute coronary syndrome (ACS) does not lead to a higher proportion of major adverse cardiac events (MACE) than the current standard process (non-inferiority design).
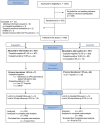
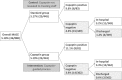
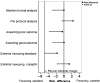
Methods and results: A total of 902 patients were randomly assigned to either standard care or the copeptin group where patients with negative troponin and copeptin values at admission were eligible for discharge after final clinical assessment. The proportion of MACE (death, survived sudden cardiac death, acute myocardial infarction (AMI), re-hospitalization for ACS, acute unplanned percutaneous coronary intervention, coronary artery bypass grafting, or documented life threatening arrhythmias) was assessed after 30 days. Intention to treat analysis showed a MACE proportion of 5.17% [95% confidence intervals (CI) 3.30-7.65%; 23/445] in the standard group and 5.19% (95% CI 3.32-7.69%; 23/443) in the copeptin group. In the per protocol analysis, the MACE proportion was 5.34% (95% CI 3.38-7.97%) in the standard group, and 3.01% (95% CI 1.51-5.33%) in the copeptin group. These results were also corroborated by sensitivity analyses. In the copeptin group, discharged copeptin negative patients had an event rate of 0.6% (2/362).
Conclusion: After clinical work-up and single combined testing of troponin and copeptin to rule-out AMI, early discharge of low- to intermediate risk patients with suspected ACS seems to be safe and has the potential to shorten length of stay in the ED. However, our results need to be confirmed in larger clinical trials or registries, before a clinical directive can be propagated.
Keywords: Acute coronary syndrome (ACS); Acute myocardial infarction (AMI); Copeptin; Randomized controlled trial (RCT); Rule-out.
© The Author 2014. Published by Oxford University Press on behalf of the European Society of Cardiology.
Figures
Comment in
-
Can copeptin emerge from the growing shadow of the troponins?Eur Heart J. 2015 Feb 7;36(6):333-6. doi: 10.1093/eurheartj/ehu211. Epub 2014 May 20. Eur Heart J. 2015. PMID: 24847154 No abstract available.
References
-
- Mockel M, Searle J, Muller R, Slagman A, Storchmann H, Oestereich P, Wyrwich W, Ale-Abaei A, Vollert JO, Koch M, Somasundaram R. Chief complaints in medical emergencies: do they relate to underlying disease and outcome? The Charite Emergency Medicine Study (CHARITEM) Eur J Emerg Med. 2013;20:103–108. - PubMed
-
- Pope JH, Aufderheide TP, Ruthazer R, Woolard RH, Feldman JA, Beshansky JR, Griffith JL, Selker HP. Missed diagnoses of acute cardiac ischemia in the emergency department. N Engl J Med. 2000;342:1163–1170. - PubMed
-
- Morgenthaler NG, Struck J, Alonso C, Bergmann A. Assay for the measurement of copeptin, a stable peptide derived from the precursor of vasopressin. Clin Chem. 2006;52:112–119. - PubMed
-
- Keller T, Tzikas S, Zeller T, Czyz E, Lillpopp L, Ojeda FM, Roth A, Bickel C, Baldus S, Sinning CR, Wild PS, Lubos E, Peetz D, Kunde J, Hartmann O, Bergmann A, Post F, Lackner KJ, Genth-Zotz S, Nicaud V, Tiret L, Munzel TF, Blankenberg S. Copeptin improves early diagnosis of acute myocardial infarction. J Am Coll Cardiol. 2010;55:2096–2106. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

