Molecular structure, function, and dynamics of clathrin-mediated membrane traffic
- PMID: 24789820
- PMCID: PMC3996469
- DOI: 10.1101/cshperspect.a016725
Molecular structure, function, and dynamics of clathrin-mediated membrane traffic
Abstract
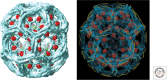
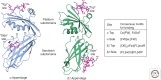
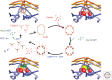
Clathrin is a molecular scaffold for vesicular uptake of cargo at the plasma membrane, where its assembly into cage-like lattices underlies the clathrin-coated pits of classical endocytosis. This review describes the structures of clathrin, major cargo adaptors, and other proteins that participate in forming a clathrin-coated pit, loading its contents, pinching off the membrane as a lattice-enclosed vesicle, and recycling the components. It integrates as much of the structural information as possible at the time of writing into a sketch of the principal steps in coated-pit and coated-vesicle formation.
Figures




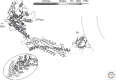

References
-
- Anderson RG, Goldstein JL, Brown MS 1977. A mutation that impairs the ability of lipoprotein receptors to localise in coated pits on the cell surface of human fibroblasts. Nature 270: 695–699 - PubMed
-
- Bao H, Daniels RW, MacLeod GT, Charlton MP, Atwood HL, Zhang B 2005. AP180 maintains the distribution of synaptic and vesicle proteins in the nerve terminal and indirectly regulates the efficacy of Ca2+-triggered exocytosis. J Neurophysiol 94: 1888–1903 - PubMed
-
- Bennett EM, Chen CY, Engqvist-Goldstein AE, Drubin DG, Brodsky FM 2001. Clathrin hub expression dissociates the actin-binding protein Hip1R from coated pits and disrupts their alignment with the actin cytoskeleton. Traffic 2: 851–858 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
