3-Hydroxyl-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor (statin)-induced 28-kDa interleukin-1β interferes with mature IL-1β signaling
- PMID: 24790079
- PMCID: PMC4047391
- DOI: 10.1074/jbc.M114.571505
3-Hydroxyl-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor (statin)-induced 28-kDa interleukin-1β interferes with mature IL-1β signaling
Abstract
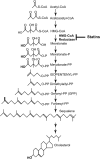
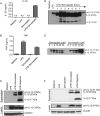
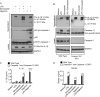
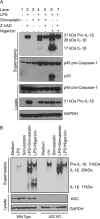
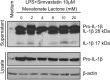
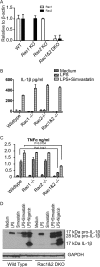
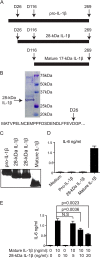
Multiple clinical trials have shown that the 3-hydroxyl-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitors known as statins have anti-inflammatory effects. However, the underlying molecular mechanism remains unclear. The proinflammatory cytokine interleukin-1β (IL-1β) is synthesized as a non-active precursor. The 31-kDa pro-IL-1β is processed into the 17-kDa active form by caspase-1-activating inflammasomes. Here, we report a novel signaling pathway induced by statins, which leads to processing of pro-IL-1β into an intermediate 28-kDa form. This statin-induced IL-1β processing is independent of caspase-1- activating inflammasomes. The 28-kDa form of IL-1β cannot activate interleukin-1 receptor-1 (IL1R1) to signal inflammatory responses. Instead, it interferes with mature IL-1β signaling through IL-1R1 and therefore may dampen inflammatory responses initiated by mature IL-1β. These results may provide new clues to explain the anti-inflammatory effects of statins.
Keywords: Atherosclerosis; Caspase; Immunology; Inflammation; Innate Immunity; Interleukin.
© 2014 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures

References
-
- Goldstein J. L., Brown M. S. (1990) Regulation of the mevalonate pathway. Nature 343, 425–430 - PubMed
-
- Downs J. R., Clearfield M., Weis S., Whitney E., Shapiro D. R., Beere P. A., Langendorfer A., Stein E. A., Kruyer W., Gotto A. M., Jr. (1998) Primary prevention of acute coronary events with lovastatin in men and women with average cholesterol levels: results of AFCAPS/TexCAPS. Air Force/Texas Coronary Atherosclerosis Prevention Study. JAMA 279, 1615–1622 - PubMed
-
- Shepherd J., Cobbe S. M., Ford I., Isles C. G., Lorimer A. R., MacFarlane P. W., McKillop J. H., Packard C. J. (1995) Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. West of Scotland Coronary Prevention Study Group. N. Engl. J. Med. 333, 1301–1307 - PubMed
-
- The Long-Term Intervention with Pravastatin in Ischaemic Disease (LIPID) Study Group (1998) Prevention of cardiovascular events and death with pravastatin in patients with coronary heart disease and a broad range of initial cholesterol levels. N. Engl. J. Med. 339, 1349–1357 - PubMed
-
- Sacks F. M., Pfeffer M. A., Moye L. A., Rouleau J. L., Rutherford J. D., Cole T. G., Brown L., Warnica J. W., Arnold J. M., Wun C. C., Davis B. R., Braunwald E. (1996) The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events Trial investigators. N. Engl. J. Med. 335, 1001–1009 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

