A quick and efficient method to generate mammalian stable cell lines based on a novel inducible alphavirus DNA/RNA layered system
- PMID: 24794511
- PMCID: PMC11113970
- DOI: 10.1007/s00018-014-1631-2
A quick and efficient method to generate mammalian stable cell lines based on a novel inducible alphavirus DNA/RNA layered system
Abstract
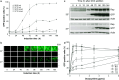
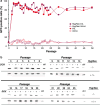
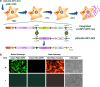
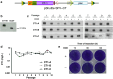
We report a new method to generate high-expressing mammalian cell lines in a quick and efficient way. For that purpose, we developed a master cell line (MCL) containing an inducible alphavirus vector expressing GFP integrated into the genome. In the MCL, recombinant RNA levels increased >4,600-fold after induction, due to a doxycycline-dependent RNA amplification loop. The MCL maintained inducibility and expression during 50 passages, being more efficient for protein expression than a conventional cell line. To generate new cell lines, mutant LoxP sites were inserted into the MCL, allowing transgene and selection gene exchange by Cre-directed recombination, leading to quick generation of inducible cell lines expressing proteins of therapeutic interest, like human cardiotrophin-1 and oncostatin-M at several mg/l/24 h. These proteins contained posttranslational modifications, showed bioactivity, and were efficiently purified. Remarkably, this system allowed production of toxic proteins, like oncostatin-M, since cells able to express it could be grown to the desired amount before induction. These cell lines were easily adapted to growth in suspension, making this methodology very attractive for therapeutic protein production.
Figures



Similar articles
-
Recent patents on alphavirus protein expression and vector production.Recent Pat Biotechnol. 2011 Dec;5(3):212-26. doi: 10.2174/187220811797579060. Recent Pat Biotechnol. 2011. PMID: 22360469 Review.
-
Development of a novel adenovirus-alphavirus hybrid vector with RNA replicon features for malignant hematopoietic cell transduction.Cancer Gene Ther. 2013 Aug;20(8):429-36. doi: 10.1038/cgt.2013.37. Epub 2013 Jul 5. Cancer Gene Ther. 2013. PMID: 23827880
-
Generation of cell lines to complement adenovirus vectors using recombination-mediated cassette exchange.BMC Biotechnol. 2010 Dec 23;10:92. doi: 10.1186/1472-6750-10-92. BMC Biotechnol. 2010. PMID: 21182761 Free PMC article.
-
Development of an all-in-one lentiviral vector system based on the original TetR for the easy generation of Tet-ON cell lines.PLoS One. 2011;6(8):e23734. doi: 10.1371/journal.pone.0023734. Epub 2011 Aug 18. PLoS One. 2011. PMID: 21876765 Free PMC article.
-
Large-scale transient expression in mammalian cells for recombinant protein production.Curr Opin Biotechnol. 1999 Apr;10(2):156-9. doi: 10.1016/s0958-1669(99)80027-5. Curr Opin Biotechnol. 1999. PMID: 10209142 Review.
Cited by
-
Enhanced therapeutic effect using sequential administration of antigenically distinct oncolytic viruses expressing oncostatin M in a Syrian hamster orthotopic pancreatic cancer model.Mol Cancer. 2015 Dec 16;14:210. doi: 10.1186/s12943-015-0479-x. Mol Cancer. 2015. PMID: 26671477 Free PMC article.
-
A Fast and Reliable Method to Generate Pure, Single Cell-derived Clones of Mammalian Cells.Bio Protoc. 2022 Aug 20;12(16):e4490. doi: 10.21769/BioProtoc.4490. eCollection 2022 Aug 20. Bio Protoc. 2022. PMID: 36505273 Free PMC article.
-
1-(7-Chloroquinolin-4-yl)-N-(4-Methoxybenzyl)-5-Methyl-1H-1,2, 3-Triazole-4- carboxamide Reduces Aβ Formation and Tau Phosphorylation in Cellular Models of Alzheimer's Disease.Neurochem Res. 2022 Apr;47(4):1110-1122. doi: 10.1007/s11064-021-03514-8. Epub 2022 Feb 15. Neurochem Res. 2022. PMID: 35165799
-
Viral and Synthetic RNA Vector Technologies and Applications.Mol Ther. 2016 Sep;24(9):1513-27. doi: 10.1038/mt.2016.143. Epub 2016 Jul 5. Mol Ther. 2016. PMID: 27377044 Free PMC article. Review.
References
-
- Pushko P, Parker M, Ludwig GV, Davis NL, Johnston RE, Smith JF. Replicon-helper systems from attenuated Venezuelan equine encephalitis virus: expression of heterologous genes in vitro and immunization against heterologous pathogens in vivo. Virology. 1997;239(2):389–401. doi: 10.1006/viro.1997.8878. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

