Functional genomic characterization of virulence factors from necrotizing fasciitis-causing strains of Aeromonas hydrophila
- PMID: 24795370
- PMCID: PMC4068668
- DOI: 10.1128/AEM.00486-14
Functional genomic characterization of virulence factors from necrotizing fasciitis-causing strains of Aeromonas hydrophila
Abstract
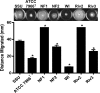
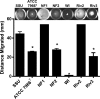
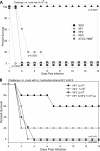
The genomes of 10 Aeromonas isolates identified and designated Aeromonas hydrophila WI, Riv3, and NF1 to NF4; A. dhakensis SSU; A. jandaei Riv2; and A. caviae NM22 and NM33 were sequenced and annotated. Isolates NF1 to NF4 were from a patient with necrotizing fasciitis (NF). Two environmental isolates (Riv2 and -3) were from the river water from which the NF patient acquired the infection. While isolates NF2 to NF4 were clonal, NF1 was genetically distinct. Outside the conserved core genomes of these 10 isolates, several unique genomic features were identified. The most virulent strains possessed one of the following four virulence factors or a combination of them: cytotoxic enterotoxin, exotoxin A, and type 3 and 6 secretion system effectors AexU and Hcp. In a septicemic-mouse model, SSU, NF1, and Riv2 were the most virulent, while NF2 was moderately virulent. These data correlated with high motility and biofilm formation by the former three isolates. Conversely, in a mouse model of intramuscular infection, NF2 was much more virulent than NF1. Isolates NF2, SSU, and Riv2 disseminated in high numbers from the muscular tissue to the visceral organs of mice, while NF1 reached the liver and spleen in relatively lower numbers on the basis of colony counting and tracking of bioluminescent strains in real time by in vivo imaging. Histopathologically, degeneration of myofibers with significant infiltration of polymorphonuclear cells due to the highly virulent strains was noted. Functional genomic analysis provided data that allowed us to correlate the highly infectious nature of Aeromonas pathotypes belonging to several different species with virulence signatures and their potential ability to cause NF.
Copyright © 2014, American Society for Microbiology. All Rights Reserved.
Figures


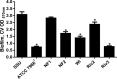





Similar articles
-
T6SS and ExoA of flesh-eating Aeromonas hydrophila in peritonitis and necrotizing fasciitis during mono- and polymicrobial infections.Proc Natl Acad Sci U S A. 2019 Nov 26;116(48):24084-24092. doi: 10.1073/pnas.1914395116. Epub 2019 Nov 11. Proc Natl Acad Sci U S A. 2019. PMID: 31712444 Free PMC article.
-
Cross-talk among flesh-eating Aeromonas hydrophila strains in mixed infection leading to necrotizing fasciitis.Proc Natl Acad Sci U S A. 2016 Jan 19;113(3):722-7. doi: 10.1073/pnas.1523817113. Epub 2016 Jan 5. Proc Natl Acad Sci U S A. 2016. PMID: 26733683 Free PMC article.
-
Characterization of Aeromonas hydrophila wound pathotypes by comparative genomic and functional analyses of virulence genes.mBio. 2013 Apr 23;4(2):e00064-13. doi: 10.1128/mBio.00064-13. mBio. 2013. PMID: 23611906 Free PMC article.
-
Characterization of virulence properties and multi-drug resistance profiles in motile Aeromonas spp. isolated from zebrafish (Danio rerio).Lett Appl Microbiol. 2018 Dec;67(6):598-605. doi: 10.1111/lam.13075. Epub 2018 Nov 12. Lett Appl Microbiol. 2018. PMID: 30229985
-
Necrotizing fasciitis and sepsis caused by Aeromonas hydrophila after crush injury of the lower extremity.Surg Infect (Larchmt). 2008 Aug;9(4):459-67. doi: 10.1089/sur.2007.028. Surg Infect (Larchmt). 2008. PMID: 18687029 Review.
Cited by
-
Crystal Structure of Exotoxin A from Aeromonas Pathogenic Species.Toxins (Basel). 2020 Jun 15;12(6):397. doi: 10.3390/toxins12060397. Toxins (Basel). 2020. PMID: 32549399 Free PMC article.
-
T6SS and ExoA of flesh-eating Aeromonas hydrophila in peritonitis and necrotizing fasciitis during mono- and polymicrobial infections.Proc Natl Acad Sci U S A. 2019 Nov 26;116(48):24084-24092. doi: 10.1073/pnas.1914395116. Epub 2019 Nov 11. Proc Natl Acad Sci U S A. 2019. PMID: 31712444 Free PMC article.
-
Multidrug-resistant Aeromonas hydrophila causing fatal bilateral necrotizing fasciitis in an immunocompromised patient: a case report.J Med Case Rep. 2018 Nov 1;12(1):326. doi: 10.1186/s13256-018-1854-1. J Med Case Rep. 2018. PMID: 30382899 Free PMC article.
-
Prediction and Analysis in silico of Genomic Islands in Aeromonas hydrophila.Front Microbiol. 2021 Nov 29;12:769380. doi: 10.3389/fmicb.2021.769380. eCollection 2021. Front Microbiol. 2021. PMID: 34912316 Free PMC article.
-
Differential production and secretion of potentially toxigenic extracellular proteins from hypervirulent Aeromonas hydrophila under biofilm and planktonic culture.BMC Microbiol. 2021 Jan 6;21(1):8. doi: 10.1186/s12866-020-02065-2. BMC Microbiol. 2021. PMID: 33407117 Free PMC article.
References
-
- Presley SM, Rainwater TR, Austin GP, Platt SG, Zak JC, Cobb GP, Marsland EJ, Tian K, Zhang B, Anderson TA, Cox SB, Abel MT, Leftwich BD, Huddleston JR, Jeter RM, Kendall RJ. 2006. Assessment of pathogens and toxicants in New Orleans, LA following Hurricane Katrina. Environ. Sci. Technol. 40:468–474. 10.1021/es052219p - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

