Acute reduction of microglia does not alter axonal injury in a mouse model of repetitive concussive traumatic brain injury
- PMID: 24797413
- PMCID: PMC4170981
- DOI: 10.1089/neu.2013.3320
Acute reduction of microglia does not alter axonal injury in a mouse model of repetitive concussive traumatic brain injury
Abstract
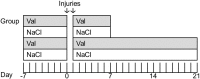
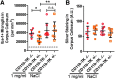
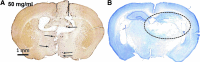
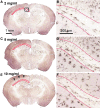
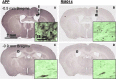
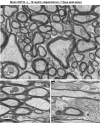
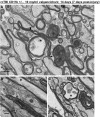
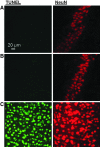
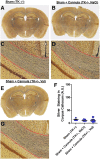
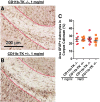
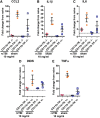
The pathological processes that lead to long-term consequences of multiple concussions are unclear. Primary mechanical damage to axons during concussion is likely to contribute to dysfunction. Secondary damage has been hypothesized to be induced or exacerbated by inflammation. The main inflammatory cells in the brain are microglia, a type of macrophage. This research sought to determine the contribution of microglia to axon degeneration after repetitive closed-skull traumatic brain injury (rcTBI) using CD11b-TK (thymidine kinase) mice, a valganciclovir-inducible model of macrophage depletion. Low-dose (1 mg/mL) valganciclovir was found to reduce the microglial population in the corpus callosum and external capsule by 35% after rcTBI in CD11b-TK mice. At both acute (7 days) and subacute (21 days) time points after rcTBI, reduction of the microglial population did not alter the extent of axon injury as visualized by silver staining. Further reduction of the microglial population by 56%, using an intermediate dose (10 mg/mL), also did not alter the extent of silver staining, amyloid precursor protein accumulation, neurofilament labeling, or axon injury evident by electron microscopy at 7 days postinjury. Longer treatment of CD11b-TK mice with intermediate dose and treatment for 14 days with high-dose (50 mg/mL) valganciclovir were both found to be toxic in this injury model. Altogether, these data are most consistent with the idea that microglia do not contribute to acute axon degeneration after multiple concussive injuries. The possibility of longer-term effects on axon structure or function cannot be ruled out. Nonetheless, alternative strategies directly targeting injury to axons may be a more beneficial approach to concussion treatment than targeting secondary processes of microglial-driven inflammation.
Keywords: axon injury; concussion; microglia.
Figures





References
-
- Corsellis J.A., Bruton C.J., and Freeman-Browne D. (1973). The aftermath of boxing. Psychol. Med. 3, 270–303 - PubMed
-
- Mortimer J.A. (1985). Epidemiology of post-traumatic encephalopathy in boxers. Minn. Med. 68, 299–300 - PubMed
-
- Geddes J.F., Vowles G.H., Nicoll J.A., and Revesz T. (1999). Neuronal cytoskeletal changes are an early consequence of repetitive head injury. Acta Neuropathol. 98, 171–178 - PubMed
-
- Guo Z, Cupples LA, Kurz A, Auerbach SH, Volicer L, Chui H, Green RC, Sadovnick AD, Duara R, DeCarli C, Johnson K, Go RC, Growdon JH, Haines JL, Kukull WA, and Farrer L.A. (2000). Head injury and the risk of AD in the MIRAGE study. Neurology 54, 1316–1323 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

