Salt stress and senescence: identification of cross-talk regulatory components
- PMID: 24803504
- PMCID: PMC4106443
- DOI: 10.1093/jxb/eru173
Salt stress and senescence: identification of cross-talk regulatory components
Abstract
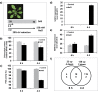
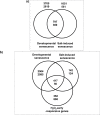
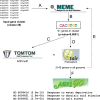
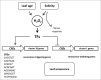
Leaf senescence is an active process with a pivotal impact on plant productivity. It results from extensive signalling cross-talk coordinating environmental factors with intrinsic age-related mechanisms. Although many studies have shown that leaf senescence is affected by a range of external parameters, knowledge about the regulatory systems that govern the interplay between developmental programmes and environmental stress is still vague. Salinity is one of the most important environmental stresses that promote leaf senescence and thus affect crop yield. Improving salt tolerance by avoiding or delaying senescence under stress will therefore play an important role in maintaining high agricultural productivity. Experimental evidence suggests that hydrogen peroxide (H2O2) functions as a common signalling molecule in both developmental and salt-induced leaf senescence. In this study, microarray-based gene expression profiling on Arabidopsis thaliana plants subjected to long-term salinity stress to induce leaf senescence was performed, together with co-expression network analysis for H2O2-responsive genes that are mutually up-regulated by salt induced- and developmental leaf senescence. Promoter analysis of tightly co-expressed genes led to the identification of seven cis-regulatory motifs, three of which were known previously, namely CACGTGT and AAGTCAA, which are associated with reactive oxygen species (ROS)-responsive genes, and CCGCGT, described as a stress-responsive regulatory motif, while the others, namely ACGCGGT, AGCMGNC, GMCACGT, and TCSTYGACG were not characterized previously. These motifs are proposed to be novel elements involved in the H2O2-mediated control of gene expression during salinity stress-triggered and developmental senescence, acting through upstream transcription factors that bind to these sites.
Keywords: Arabidopsis; hydrogen peroxide; longevity; reactive oxygen species; salt stress; senescence; signal cross-talk; transcription factor..
© The Author 2014. Published by Oxford University Press on behalf of the Society for Experimental Biology.
Figures

References
-
- Ashraf M. 2010. Inducing drought tolerance in plants: some recent advances. Biotechnology Advances 28, 169–183 - PubMed
-
- Bailey T, Elkan C. 1994. Fitting a mixture model by expectation maximization to discover motifs in biopolymers. Proceedings of the International Conference on Intelligent Systems for Molecular Biology 2, 28–36 - PubMed
-
- Bailey TL, Elkan C. 1995. The value of prior knowledge in discovering motifs with MEME. Proceedings of the Third International Conference on Intelligent Systems for Molecular Biology. Palo Alto, CA: AAAI Press, 21–29 - PubMed
-
- Balazadeh S, Rianño-Pachon DM, Mueller-Roeber B. 2008. Transcription factors regulating leaf senescence in Arabidopsis thaliana . Plant Biology 10 (Suppl. 1), 63–75 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

