Lingual muscle activity across sleep-wake States in rats with surgically altered upper airway
- PMID: 24803913
- PMCID: PMC4009435
- DOI: 10.3389/fneur.2014.00061
Lingual muscle activity across sleep-wake States in rats with surgically altered upper airway
Abstract
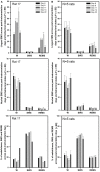
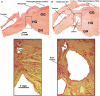
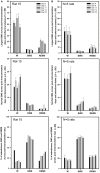
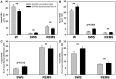
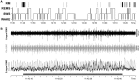
Obstructive sleep apnea (OSA) patients have increased upper airway muscle activity, including such lingual muscles as the genioglossus (GG), geniohyoid (GH), and hyoglossus (HG). This adaptation partially protects their upper airway against obstructions. Rodents are used to study the central neural control of sleep and breathing but they do not naturally exhibit OSA. We investigated whether, in chronically instrumented, behaving rats, disconnecting the GH and HG muscles from the hyoid (H) apparatus would result in a compensatory increase of other upper airway muscle activity (electromyogram, EMG) and/or other signs of upper airway instability. We first determined that, in intact rats, lingual (GG and intrinsic) muscles maintained stable activity levels when quantified based on 2 h-long recordings conducted on days 6 through 22 after instrumentation. We then studied five rats in which the tendons connecting the GH and HG muscles to the H apparatus were experimentally severed. When quantified across all recording days, lingual EMG during slow-wave sleep (SWS) was modestly but significantly increased in rats with surgically altered upper airway [8.6 ± 0.7% (SE) vs. 6.1 ± 0.7% of the mean during wakefulness; p = 0.012]. Respiratory modulation of lingual EMG occurred mainly during SWS and was similarly infrequent in both groups, and the incidence of sighs and central apneas also was similar. Thus, a weakened action of selected lingual muscles did not produce sleep-disordered breathing but resulted in a relatively elevated activity in other lingual muscles during SWS. These results encourage more extensive surgical manipulations with the aim to obtain a rodent model with collapsible upper airway.
Keywords: REM sleep; genioglossus; hyoid bone; obstructive sleep apnea; tongue.
Figures



Similar articles
-
Neural Control of the Upper Airway: Respiratory and State-Dependent Mechanisms.Compr Physiol. 2016 Sep 15;6(4):1801-1850. doi: 10.1002/cphy.c160002. Compr Physiol. 2016. PMID: 27783860 Free PMC article. Review.
-
Electromyographic activity at the base and tip of the tongue across sleep-wake states in rats.Respir Physiol Neurobiol. 2009 Jul 31;167(3):307-15. doi: 10.1016/j.resp.2009.06.004. Epub 2009 Jun 17. Respir Physiol Neurobiol. 2009. PMID: 19539786 Free PMC article.
-
Respiratory modulation of lingual muscle activity across sleep-wake states in rats.Respir Physiol Neurobiol. 2013 Sep 15;188(3):308-17. doi: 10.1016/j.resp.2013.05.033. Epub 2013 May 31. Respir Physiol Neurobiol. 2013. PMID: 23732510 Free PMC article.
-
Differential effect of sleep-wake states on lingual and dorsal neck muscle activity in rats.Respir Physiol Neurobiol. 2005 Jul 28;147(2-3):191-203. doi: 10.1016/j.resp.2005.05.007. Respir Physiol Neurobiol. 2005. PMID: 15964252
-
Sleeping tongue: current perspectives of genioglossus control in healthy individuals and patients with obstructive sleep apnea.Nat Sci Sleep. 2018 Jun 15;10:169-179. doi: 10.2147/NSS.S143296. eCollection 2018. Nat Sci Sleep. 2018. PMID: 29942169 Free PMC article. Review.
Cited by
-
Hypoglossal motoneurons are endogenously activated by serotonin during the active period of circadian cycle.Respir Physiol Neurobiol. 2018 Jan;248:17-24. doi: 10.1016/j.resp.2017.11.002. Epub 2017 Nov 9. Respir Physiol Neurobiol. 2018. PMID: 29129751 Free PMC article.
-
Neural Control of the Upper Airway: Respiratory and State-Dependent Mechanisms.Compr Physiol. 2016 Sep 15;6(4):1801-1850. doi: 10.1002/cphy.c160002. Compr Physiol. 2016. PMID: 27783860 Free PMC article. Review.
-
Catecholaminergic A1/C1 neurons contribute to the maintenance of upper airway muscle tone but may not participate in NREM sleep-related depression of these muscles.Respir Physiol Neurobiol. 2017 Oct;244:41-50. doi: 10.1016/j.resp.2017.07.001. Epub 2017 Jul 12. Respir Physiol Neurobiol. 2017. PMID: 28711601 Free PMC article.
-
Effects of Cannabinoid Agonists and Antagonists on Sleep and Breathing in Sprague-Dawley Rats.Sleep. 2017 Sep 1;40(9):zsx112. doi: 10.1093/sleep/zsx112. Sleep. 2017. PMID: 28934522 Free PMC article.
-
Modulation of Motoneuronal Activity With Sleep-Wake States and Motoneuronal Gene Expression Vary With Circadian Rest-Activity Cycle.Front Integr Neurosci. 2018 Aug 7;12:32. doi: 10.3389/fnint.2018.00032. eCollection 2018. Front Integr Neurosci. 2018. PMID: 30131680 Free PMC article.
References
-
- Kubin L, Davies RO. Mechanisms of upper airway hypotonia. 2nd ed In: Pack AI, editor. Sleep Apnea: Pathogenesis, Diagnosis, and Treatment. St. Helier: Informa Healthcare; (2011). p. 82–127
-
- Remmers JE, DeGroot WJ, Sauerland EK, Anch AM. Pathogenesis of upper airway occlusion during sleep. J Appl Physiol (1978) 44:931–8 - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

