Co-expression of foreign proteins tethered to HIV-1 envelope glycoprotein on the cell surface by introducing an intervening second membrane-spanning domain
- PMID: 24804933
- PMCID: PMC4013048
- DOI: 10.1371/journal.pone.0096790
Co-expression of foreign proteins tethered to HIV-1 envelope glycoprotein on the cell surface by introducing an intervening second membrane-spanning domain
Abstract
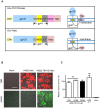
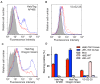
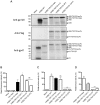
The envelope glycoprotein (Env) of human immunodeficiency virus type I (HIV-1) mediates membrane fusion. To analyze the mechanism of HIV-1 Env-mediated membrane fusion, it is desirable to determine the expression level of Env on the cell surface. However, the quantification of Env by immunological staining is often hampered by the diversity of HIV-1 Env and limited availability of universal antibodies that recognize different Envs with equal efficiency. To overcome this problem, here we linked a tag protein called HaloTag at the C-terminus of HIV-1 Env. To relocate HaloTag to the cell surface, we introduced a second membrane-spanning domain (MSD) between Env and HaloTag. The MSD of transmembrane protease serine 11D, a type II transmembrane protein, successfully relocated HaloTag to the cell surface. The surface level of Env can be estimated indirectly by staining HaloTag with a specific membrane-impermeable fluorescent ligand. This tagging did not compromise the fusogenicity of Env drastically. Furthermore, fusogenicity of Env was preserved even after the labeling with the ligands. We have also found that an additional foreign peptide or protein such as C34 or neutralizing single-chain variable fragment (scFv) can be linked to the C-terminus of the HaloTag protein. Using these constructs, we were able to determine the required length of C34 and critical residues of neutralizing scFv for blocking membrane fusion, respectively.
Conflict of interest statement
Figures
Similar articles
-
Role of the fusion peptide and membrane-proximal domain in HIV-1 envelope glycoprotein-mediated membrane fusion.Biochemistry. 2003 Dec 9;42(48):14150-8. doi: 10.1021/bi035154g. Biochemistry. 2003. PMID: 14640682
-
Role of envelope processing and gp41 membrane spanning domain in the formation of human immunodeficiency virus type 1 (HIV-1) fusion-competent envelope glycoprotein complex.Virus Res. 2007 Mar;124(1-2):103-12. doi: 10.1016/j.virusres.2006.10.009. Epub 2006 Nov 28. Virus Res. 2007. PMID: 17129629
-
Membrane topology analysis of HIV-1 envelope glycoprotein gp41.Retrovirology. 2010 Nov 30;7:100. doi: 10.1186/1742-4690-7-100. Retrovirology. 2010. PMID: 21118523 Free PMC article.
-
Regulation of human immunodeficiency virus type 1 envelope glycoprotein fusion by a membrane-interactive domain in the gp41 cytoplasmic tail.J Virol. 2005 Oct;79(19):12231-41. doi: 10.1128/JVI.79.19.12231-12241.2005. J Virol. 2005. PMID: 16160149 Free PMC article.
-
HIV-1 gp41 Residues Modulate CD4-Induced Conformational Changes in the Envelope Glycoprotein and Evolution of a Relaxed Conformation of gp120.J Virol. 2018 Jul 31;92(16):e00583-18. doi: 10.1128/JVI.00583-18. Print 2018 Aug 15. J Virol. 2018. PMID: 29875245 Free PMC article.
Cited by
-
Using a split luciferase assay (SLA) to measure the kinetics of cell-cell fusion mediated by herpes simplex virus glycoproteins.Methods. 2015 Nov 15;90:68-75. doi: 10.1016/j.ymeth.2015.05.021. Epub 2015 May 26. Methods. 2015. PMID: 26022509 Free PMC article. Review.
-
Discovery of New Potent anti-MERS CoV Fusion Inhibitors.Front Pharmacol. 2021 Jun 2;12:685161. doi: 10.3389/fphar.2021.685161. eCollection 2021. Front Pharmacol. 2021. PMID: 34149429 Free PMC article.
-
CADM1 and CADM2 Trigger Neuropathogenic Measles Virus-Mediated Membrane Fusion by Acting in cis.J Virol. 2021 Jun 24;95(14):e0052821. doi: 10.1128/JVI.00528-21. Epub 2021 Jun 24. J Virol. 2021. PMID: 33910952 Free PMC article.
-
Discovery of New Fusion Inhibitor Peptides against SARS-CoV-2 by Targeting the Spike S2 Subunit.Biomol Ther (Seoul). 2021 May 1;29(3):282-289. doi: 10.4062/biomolther.2020.201. Biomol Ther (Seoul). 2021. PMID: 33424013 Free PMC article.
-
Amino acid changes accumulated in the fusion protein allow neuropathogenic measles viruses to use a broad repertoire of host factors for cell fusion triggering.J Virol. 2025 May 20;99(5):e0230724. doi: 10.1128/jvi.02307-24. Epub 2025 Apr 7. J Virol. 2025. PMID: 40192292 Free PMC article.
References
-
- Dettenhofer M, Yu XF (2001) Characterization of the biosynthesis of human immunodeficiency virus type 1 Env from infected T-cells and the effects of glucose trimming of Env on virion infectivity. J Biol Chem 276: 5985–5991. - PubMed
-
- Dash B, McIntosh A, Barrett W, Daniels R (1994) Deletion of a single N-linked glycosylation site from the transmembrane envelope protein of human immunodeficiency virus type 1 stops cleavage and transport of gp160 preventing env-mediated fusion. J Gen Virol 75 (Pt 6): 1389–1397. - PubMed
-
- Wyatt R, Sodroski J (1998) The HIV-1 envelope glycoproteins: fusogens, antigens, and immunogens. Science 280: 1884–1888. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

