Activation and repression of Epstein-Barr Virus and Kaposi's sarcoma-associated herpesvirus lytic cycles by short- and medium-chain fatty acids
- PMID: 24807711
- PMCID: PMC4097796
- DOI: 10.1128/JVI.00722-14
Activation and repression of Epstein-Barr Virus and Kaposi's sarcoma-associated herpesvirus lytic cycles by short- and medium-chain fatty acids
Abstract
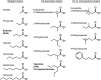
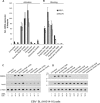
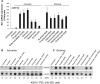
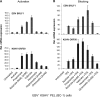
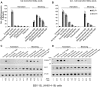
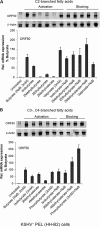
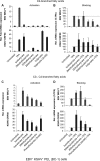
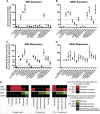
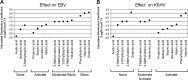
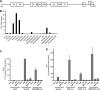
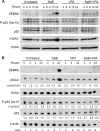
The lytic cycles of Epstein-Barr virus (EBV) and Kaposi's sarcoma-associated herpesvirus (KSHV) are induced in cell culture by sodium butyrate (NaB), a short-chain fatty acid (SCFA) histone deacetylase (HDAC) inhibitor. Valproic acid (VPA), another SCFA and an HDAC inhibitor, induces the lytic cycle of KSHV but blocks EBV lytic reactivation. To explore the hypothesis that structural differences between NaB and VPA account for their functional effects on the two related viruses, we investigated the capacity of 16 structurally related short- and medium-chain fatty acids to promote or prevent lytic cycle reactivation. SCFAs differentially affected EBV and KSHV reactivation. KSHV was reactivated by all SCFAs that are HDAC inhibitors, including phenylbutyrate. However, several fatty acid HDAC inhibitors, such as isobutyrate and phenylbutyrate, did not reactivate EBV. Reactivation of KSHV lytic transcripts could not be blocked completely by any fatty acid tested. In contrast, several medium-chain fatty acids inhibited lytic activation of EBV. Fatty acids that blocked EBV reactivation were more lipophilic than those that activated EBV. VPA blocked activation of the BZLF1 promoter by NaB but did not block the transcriptional function of ZEBRA. VPA also blocked activation of the DNA damage response that accompanies EBV lytic cycle activation. Properties of SCFAs in addition to their effects on chromatin are likely to explain activation or repression of EBV. We concluded that fatty acids stimulate the two related human gammaherpesviruses to enter the lytic cycle through different pathways. Importance: Lytic reactivation of EBV and KSHV is needed for persistence of these viruses and plays a role in carcinogenesis. Our direct comparison highlights the mechanistic differences in lytic reactivation between related human oncogenic gammaherpesviruses. Our findings have therapeutic implications, as fatty acids are found in the diet and produced by the human microbiota. Small-molecule inducers of the lytic cycle are desired for oncolytic therapy. Inhibition of viral reactivation, alternatively, may prove useful in cancer treatment. Overall, our findings contribute to the understanding of pathways that control the latent-to-lytic switch and identify naturally occurring molecules that may regulate this process.
Copyright © 2014, American Society for Microbiology. All Rights Reserved.
Figures
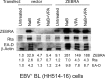
Similar articles
-
De novo protein synthesis is required for lytic cycle reactivation of Epstein-Barr virus, but not Kaposi's sarcoma-associated herpesvirus, in response to histone deacetylase inhibitors and protein kinase C agonists.J Virol. 2007 Sep;81(17):9279-91. doi: 10.1128/JVI.00982-07. Epub 2007 Jun 27. J Virol. 2007. PMID: 17596302 Free PMC article.
-
Valpromide Inhibits Lytic Cycle Reactivation of Epstein-Barr Virus.mBio. 2016 Mar 1;7(2):e00113. doi: 10.1128/mBio.00113-16. mBio. 2016. PMID: 26933051 Free PMC article.
-
Valproic acid antagonizes the capacity of other histone deacetylase inhibitors to activate the Epstein-barr virus lytic cycle.J Virol. 2011 Jun;85(11):5628-43. doi: 10.1128/JVI.02659-10. Epub 2011 Mar 16. J Virol. 2011. PMID: 21411522 Free PMC article.
-
Lytic cycle switches of oncogenic human gammaherpesviruses.Adv Cancer Res. 2007;97:81-109. doi: 10.1016/S0065-230X(06)97004-3. Adv Cancer Res. 2007. PMID: 17419942 Review.
-
EBV and KSHV Infection Dysregulates Autophagy to Optimize Viral Replication, Prevent Immune Recognition and Promote Tumorigenesis.Viruses. 2018 Oct 31;10(11):599. doi: 10.3390/v10110599. Viruses. 2018. PMID: 30384495 Free PMC article. Review.
Cited by
-
Impact of the Microbiota on Viral Infections.Annu Rev Virol. 2023 Sep 29;10(1):371-395. doi: 10.1146/annurev-virology-111821-115754. Epub 2023 Apr 18. Annu Rev Virol. 2023. PMID: 37071931 Free PMC article. Review.
-
The Fight against the Carcinogenic Epstein-Barr Virus: Gut Microbiota, Natural Medicines, and Beyond.Int J Mol Sci. 2023 Jan 15;24(2):1716. doi: 10.3390/ijms24021716. Int J Mol Sci. 2023. PMID: 36675232 Free PMC article. Review.
-
Signatures of oral microbiome in HIV-infected individuals with oral Kaposi's sarcoma and cell-associated KSHV DNA.PLoS Pathog. 2020 Jan 17;16(1):e1008114. doi: 10.1371/journal.ppat.1008114. eCollection 2020 Jan. PLoS Pathog. 2020. PMID: 31951641 Free PMC article. Clinical Trial.
-
Recent advances in the study of Kaposi's sarcoma-associated herpesvirus replication and pathogenesis.Virol Sin. 2015 Apr;30(2):130-45. doi: 10.1007/s12250-015-3595-2. Epub 2015 Apr 23. Virol Sin. 2015. PMID: 25924994 Free PMC article. Review.
-
Fatty acids role in multiple sclerosis as "metabokines".J Neuroinflammation. 2022 Jun 17;19(1):157. doi: 10.1186/s12974-022-02502-1. J Neuroinflammation. 2022. PMID: 35715809 Free PMC article. Review.
References
-
- Ma SD, Hegde S, Young KH, Sullivan R, Rajesh D, Zhou Y, Jankowska-Gan E, Burlingham WJ, Sun X, Gulley ML, Tang W, Gumperz JE, Kenney SC. 2011. A new model of Epstein-Barr virus infection reveals an important role for early lytic viral protein expression in the development of lymphomas. J. Virol. 85:165–177. 10.1128/JVI.01512-10 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

