Applications of density functional theory to iron-containing molecules of bioinorganic interest
- PMID: 24809043
- PMCID: PMC4010748
- DOI: 10.3389/fchem.2014.00014
Applications of density functional theory to iron-containing molecules of bioinorganic interest
Abstract
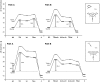
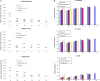
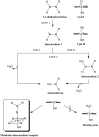
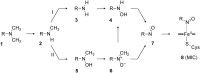
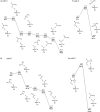
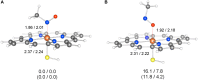
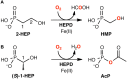
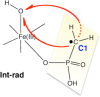
The past decades have seen an explosive growth in the application of density functional theory (DFT) methods to molecular systems that are of interest in a variety of scientific fields. Owing to its balanced accuracy and efficiency, DFT plays particularly useful roles in the theoretical investigation of large molecules. Even for biological molecules such as proteins, DFT finds application in the form of, e.g., hybrid quantum mechanics and molecular mechanics (QM/MM), in which DFT may be used as a QM method to describe a higher prioritized region in the system, while a MM force field may be used to describe remaining atoms. Iron-containing molecules are particularly important targets of DFT calculations. From the viewpoint of chemistry, this is mainly because iron is abundant on earth, iron plays powerful (and often enigmatic) roles in enzyme catalysis, and iron thus has the great potential for biomimetic catalysis of chemically difficult transformations. In this paper, we present a brief overview of several recent applications of DFT to iron-containing non-heme synthetic complexes, heme-type cytochrome P450 enzymes, and non-heme iron enzymes, all of which are of particular interest in the field of bioinorganic chemistry. Emphasis will be placed on our own work.
Keywords: QMMM; catalysis; density functional theory; enzyme reactions; iron-containing molecules; protein environment.
Figures


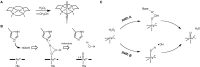




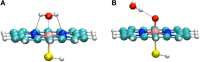



References
-
- ADF2012.01. (2012). SCM Theoretical Chemistry. Amsterdam: Vrije Universiteit; Available online at: http://www.scm.com
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

