Cab45S inhibits the ER stress-induced IRE1-JNK pathway and apoptosis via GRP78/BiP
- PMID: 24810055
- PMCID: PMC4047922
- DOI: 10.1038/cddis.2014.193
Cab45S inhibits the ER stress-induced IRE1-JNK pathway and apoptosis via GRP78/BiP
Abstract
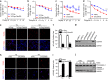
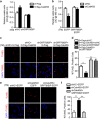
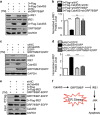
Disturbance of endoplasmic reticulum (ER) homeostasis causes ER stress and leads to activation of the unfolded protein response, which reduces the stress and promotes cell survival at the early stage of stress, or triggers cell death and apoptosis when homeostasis is not restored under prolonged ER stress. Here, we report that Cab45S, a member of the CREC family, inhibits ER stress-induced apoptosis. Depletion of Cab45S increases inositol-requiring kinase 1 (IRE1) activity, thus producing more spliced forms of X-box-binding protein 1 mRNA at the early stage of stress and leads to phosphorylation of c-Jun N-terminal kinase, which finally induces apoptosis. Furthermore, we find that Cab45S specifically interacts with 78-kDa glucose-regulated protein/immunoglobulin heavy chain binding protein (GRP78/BiP) on its nucleotide-binding domain. Cab45S enhances GRP78/BiP protein level and stabilizes the interaction of GRP78/BiP with IRE1 to inhibit ER stress-induced IRE1 activation and apoptosis. Together, Cab45S, a novel regulator of GRP78/BiP, suppresses ER stress-induced IRE1 activation and apoptosis by binding to and elevating GRP78/BiP, and has a role in the inhibition of ER stress-induced apoptosis.
Figures



Similar articles
-
Protective role of Gipie, a Girdin family protein, in endoplasmic reticulum stress responses in endothelial cells.Mol Biol Cell. 2011 Mar 15;22(6):736-47. doi: 10.1091/mbc.E10-08-0724. Epub 2011 Feb 2. Mol Biol Cell. 2011. PMID: 21289099 Free PMC article.
-
Ginsenoside Rb1 reduced ischemic stroke-induced apoptosis through endoplasmic reticulum stress-associated IRE1/TRAF2/JNK pathway.Naunyn Schmiedebergs Arch Pharmacol. 2025 Jan;398(1):747-764. doi: 10.1007/s00210-024-03292-4. Epub 2024 Jul 25. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 39052059
-
Naphthoquinone derivative PPE8 induces endoplasmic reticulum stress in p53 null H1299 cells.Oxid Med Cell Longev. 2015;2015:453679. doi: 10.1155/2015/453679. Epub 2015 Jan 18. Oxid Med Cell Longev. 2015. PMID: 25685256 Free PMC article.
-
Molecular signal networks and regulating mechanisms of the unfolded protein response.J Zhejiang Univ Sci B. 2017 Jan.;18(1):1-14. doi: 10.1631/jzus.B1600043. J Zhejiang Univ Sci B. 2017. PMID: 28070992 Free PMC article. Review.
-
Multiple Ways for Stress Sensing and Regulation of the Endoplasmic Reticulum-stress Sensors.Cell Struct Funct. 2021 May 22;46(1):37-49. doi: 10.1247/csf.21015. Epub 2021 Mar 26. Cell Struct Funct. 2021. PMID: 33775971 Free PMC article. Review.
Cited by
-
The Reciprocal Causation of the ASK1-JNK1/2 Pathway and Endoplasmic Reticulum Stress in Diabetes-Induced Cognitive Decline.Front Cell Dev Biol. 2020 Jul 17;8:602. doi: 10.3389/fcell.2020.00602. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 32766246 Free PMC article.
-
Cab45S promotes cell proliferation through SERCA2b inhibition and Ca2+ signaling.Oncogene. 2016 Jan 7;35(1):35-46. doi: 10.1038/onc.2015.56. Epub 2015 Mar 16. Oncogene. 2016. PMID: 25772237
-
New Frontiers on ER Stress Modulation: Are TRP Channels the Leading Actors?Int J Mol Sci. 2022 Dec 22;24(1):185. doi: 10.3390/ijms24010185. Int J Mol Sci. 2022. PMID: 36613628 Free PMC article. Review.
-
ATP citrate lyase inhibitor triggers endoplasmic reticulum stress to induce hepatocellular carcinoma cell apoptosis via p-eIF2α/ATF4/CHOP axis.J Cell Mol Med. 2021 Feb;25(3):1468-1479. doi: 10.1111/jcmm.16235. Epub 2021 Jan 3. J Cell Mol Med. 2021. PMID: 33393219 Free PMC article.
-
A Novel Role of Cab45-G in Mediating Cell Migration in Cancer Cells.Int J Biol Sci. 2016 Apr 25;12(6):677-87. doi: 10.7150/ijbs.11037. eCollection 2016. Int J Biol Sci. 2016. PMID: 27194945 Free PMC article.
References
-
- Pizzo P, Pozzan T. Mitochondria-endoplasmic reticulum choreography: structure and signaling dynamics. Trends Cell Biol. 2007;17:511–517. - PubMed
-
- Kim I, Xu W, Reed JC. Cell death and endoplasmic reticulum stress: disease relevance and therapeutic opportunities. Nat Rev Drug Discov. 2008;7:1013–1030. - PubMed
-
- Ron D, Walter P. Signal integration in the endoplasmic reticulum unfolded protein response. Nat Rev Mol Cell Biol. 2007;8:519–529. - PubMed
-
- Hetz C. The unfolded protein response: controlling cell fate decisions under ER stress and beyond. Nat Rev Mol Cell Biol. 2012;13:89–102. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

