Augmented osteogenic responses in human aortic valve cells exposed to oxLDL and TLR4 agonist: a mechanistic role of Notch1 and NF-κB interaction
- PMID: 24810405
- PMCID: PMC4014478
- DOI: 10.1371/journal.pone.0095400
Augmented osteogenic responses in human aortic valve cells exposed to oxLDL and TLR4 agonist: a mechanistic role of Notch1 and NF-κB interaction
Abstract
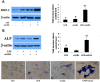
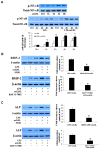
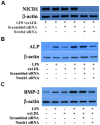
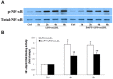
Aortic valve calcification causes the progression of calcific aortic valve disease (CAVD). Stimulation of aortic valve interstitial cells (AVICs) with lipopolysaccharide (LPS) up-regulates the expression of osteogenic mediators, and NF-κB plays a central role in mediating AVIC osteogenic responses to Toll-like receptor 4 (TLR4) stimulation. Diseased aortic valves exhibit greater levels of oxidized low-density lipoprotein (oxLDL). This study tested the hypothesis that oxLDL augments the osteogenic responses in human AVICs through modulation of NF-κB and Notch1 activation. AVICs isolated from normal human aortic valves were treated with LPS (0.1 µg/ml), oxLDL (20 µg/ml) or LPS plus oxLDL for 48 h. OxLDL alone increased cellular bone morphogenetic protein-2 (BMP-2) levels while it had no effect on alkaline phosphatase (ALP) levels. Cells exposed to LPS plus oxLDL produced higher levels of BMP-2 and ALP than cells exposed to LPS alone. Further, LPS plus oxLDL induced greater NF-κB activation, and inhibition of NF-κB markedly reduced the expression of BMP-2 and ALP in cells treated with LPS plus oxLDL. OxLDL also induced Notch1 activation and resulted in augmented Notch1 activation when it was combined with LPS. Inhibition of Notch1 cleavage attenuated NF-κB activation induced by LPS plus oxLDL, and inhibition of NF-κB suppressed the expression of BMP-2 and ALP induced by the synergistic effect of Jagged1 and LPS. These findings demonstrate that oxLDL up-regulates BMP-2 expression in human AVICs and synergizes with LPS to elicit augmented AVIC osteogenic responses. OxLDL exerts its effect through modulation of the Notch1-NF-κB signaling cascade. Thus, oxLDL may play a role in the mechanism underlying CAVD progression.
Conflict of interest statement
Figures


Similar articles
-
Activation of TLR3 induces osteogenic responses in human aortic valve interstitial cells through the NF-κB and ERK1/2 pathways.Int J Biol Sci. 2015 Mar 20;11(4):482-93. doi: 10.7150/ijbs.10905. eCollection 2015. Int J Biol Sci. 2015. PMID: 25798067 Free PMC article.
-
Notch1 promotes the pro-osteogenic response of human aortic valve interstitial cells via modulation of ERK1/2 and nuclear factor-κB activation.Arterioscler Thromb Vasc Biol. 2013 Jul;33(7):1580-90. doi: 10.1161/ATVBAHA.112.300912. Epub 2013 May 2. Arterioscler Thromb Vasc Biol. 2013. PMID: 23640488 Free PMC article.
-
Interleukin-37 suppresses the osteogenic responses of human aortic valve interstitial cells in vitro and alleviates valve lesions in mice.Proc Natl Acad Sci U S A. 2017 Feb 14;114(7):1631-1636. doi: 10.1073/pnas.1619667114. Epub 2017 Jan 30. Proc Natl Acad Sci U S A. 2017. PMID: 28137840 Free PMC article.
-
Molecular and cellular aspects of calcific aortic valve disease.Circ Res. 2013 Jul 5;113(2):198-208. doi: 10.1161/CIRCRESAHA.113.300155. Circ Res. 2013. PMID: 23833294 Free PMC article. Review.
-
A Notch more: Molecular players in bicuspid aortic valve disease.J Mol Cell Cardiol. 2019 Sep;134:62-68. doi: 10.1016/j.yjmcc.2019.05.018. Epub 2019 May 28. J Mol Cell Cardiol. 2019. PMID: 31150732 Review.
Cited by
-
Inflammation and Mechanical Stress Stimulate Osteogenic Differentiation of Human Aortic Valve Interstitial Cells.Front Physiol. 2018 Nov 20;9:1635. doi: 10.3389/fphys.2018.01635. eCollection 2018. Front Physiol. 2018. PMID: 30524301 Free PMC article.
-
Creation of disease-inspired biomaterial environments to mimic pathological events in early calcific aortic valve disease.Proc Natl Acad Sci U S A. 2018 Jan 16;115(3):E363-E371. doi: 10.1073/pnas.1704637115. Epub 2017 Dec 27. Proc Natl Acad Sci U S A. 2018. PMID: 29282325 Free PMC article.
-
Biomolecules Orchestrating Cardiovascular Calcification.Biomolecules. 2021 Oct 7;11(10):1482. doi: 10.3390/biom11101482. Biomolecules. 2021. PMID: 34680115 Free PMC article. Review.
-
Macrophages in Calcific Aortic Valve Disease: Paracrine and Juxtacrine Disease Drivers.Biomolecules. 2024 Dec 2;14(12):1547. doi: 10.3390/biom14121547. Biomolecules. 2024. PMID: 39766254 Free PMC article. Review.
-
Activation of TLR3 induces osteogenic responses in human aortic valve interstitial cells through the NF-κB and ERK1/2 pathways.Int J Biol Sci. 2015 Mar 20;11(4):482-93. doi: 10.7150/ijbs.10905. eCollection 2015. Int J Biol Sci. 2015. PMID: 25798067 Free PMC article.
References
-
- Mohler ER III (2004) Mechanisms of aortic valve calcification. Am J Cardiol 94: 1396–1402, A1396. - PubMed
-
- Messika-Zeitoun D, Bielak LF, Peyser PA, Sheedy PF, Turner ST, et al. (2007) Aortic valve calcification: determinants and progression in the population. Arterioscler Thromb Vasc Biol 27: 642–648. - PubMed
-
- Osman L, Yacoub MH, Latif N, Amrani M, Chester AH (2006) Role of human valve interstitial cells in valve calcification and their response to atorvastatin. Circulation 114: I547–552. - PubMed
-
- Meng X, Ao L, Song Y, Babu A, Yang X, et al. (2008) Expression of functional Toll-like receptors 2 and 4 in human aortic valve interstitial cells: potential roles in aortic valve inflammation and stenosis. Am J Physiol Cell Physiol 294: C29–C35. - PubMed
-
- Yang X, Fullerton DA, Su X, Ao L, Cleveland JC, et al. (2009) Pro-osteogenic phenotype of human aortic valve interstitial cells is associated with higher levels of Toll-like receptors 2 and 4 and enhanced expression of bone morphogenetic protein 2. J Am Coll Cardiol 53: 491–500. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

