Autophagy promotes radiation-induced senescence but inhibits bystander effects in human breast cancer cells
- PMID: 24813621
- PMCID: PMC4203548
- DOI: 10.4161/auto.28772
Autophagy promotes radiation-induced senescence but inhibits bystander effects in human breast cancer cells
Abstract
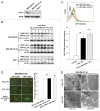
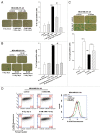
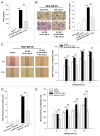
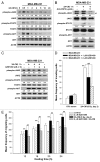
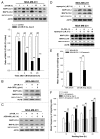
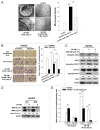
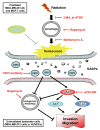
Ionizing radiation induces cellular senescence to suppress cancer cell proliferation. However, it also induces deleterious bystander effects in the unirradiated neighboring cells through the release of senescence-associated secretory phenotypes (SASPs) that promote tumor progression. Although autophagy has been reported to promote senescence, its role is still unclear. We previously showed that radiation induces senescence in PTTG1-depleted cancer cells. In this study, we found that autophagy was required for the radiation-induced senescence in PTTG1-depleted breast cancer cells. Inhibition of autophagy caused the cells to switch from radiation-induced senescence to apoptosis. Senescent cancer cells exerted bystander effects by promoting the invasion and migration of unirradiated cells through the release of CSF2 and the subsequently activation of the JAK2-STAT3 and AKT pathways. However, the radiation-induced bystander effects were correlated with the inhibition of endogenous autophagy in bystander cells, which also resulted from the activation of the CSF2-JAK2 pathway. The induction of autophagy by rapamycin reduced the radiation-induced bystander effects. This study reveals, for the first time, the dual role of autophagy in radiation-induced senescence and bystander effects.
Keywords: angiogenesis; autophagy; invasion; migration; radiation; senescence-associated secretory phenotypes.
Figures


Similar articles
-
Radiation induces senescence and a bystander effect through metabolic alterations.Cell Death Dis. 2014 May 22;5(5):e1255. doi: 10.1038/cddis.2014.220. Cell Death Dis. 2014. PMID: 24853433 Free PMC article.
-
Radiation-induced senescence in securin-deficient cancer cells promotes cell invasion involving the IL-6/STAT3 and PDGF-BB/PDGFR pathways.Sci Rep. 2013;3:1675. doi: 10.1038/srep01675. Sci Rep. 2013. PMID: 23591770 Free PMC article.
-
Ginsenoside Rh2 Ameliorates Doxorubicin-Induced Senescence Bystander Effect in Breast Carcinoma Cell MDA-MB-231 and Normal Epithelial Cell MCF-10A.Int J Mol Sci. 2019 Mar 12;20(5):1244. doi: 10.3390/ijms20051244. Int J Mol Sci. 2019. PMID: 30871042 Free PMC article.
-
The Roles of Autophagy and Senescence in the Tumor Cell Response to Radiation.Radiat Res. 2020 Aug 1;194(2):103-115. doi: 10.1667/RADE-20-00009. Radiat Res. 2020. PMID: 32845995 Free PMC article. Review.
-
Ionizing radiation-induced bystander effects, potential targets for modulation of radiotherapy.Eur J Pharmacol. 2009 Dec 25;625(1-3):156-64. doi: 10.1016/j.ejphar.2009.07.028. Epub 2009 Oct 14. Eur J Pharmacol. 2009. PMID: 19835860 Review.
Cited by
-
Knock-Down of Mucolipin 1 Channel Promotes Tumor Progression and Invasion in Human Glioblastoma Cell Lines.Front Oncol. 2021 Apr 19;11:578928. doi: 10.3389/fonc.2021.578928. eCollection 2021. Front Oncol. 2021. PMID: 33954107 Free PMC article.
-
Targeted and Non-Targeted Mechanisms for Killing Hypoxic Tumour Cells-Are There New Avenues for Treatment?Int J Mol Sci. 2021 Aug 11;22(16):8651. doi: 10.3390/ijms22168651. Int J Mol Sci. 2021. PMID: 34445354 Free PMC article. Review.
-
Autophagy Is Pro-Senescence When Seen in Close-Up, but Anti-Senescence in Long-Shot.Mol Cells. 2017 Sep 30;40(9):607-612. doi: 10.14348/molcells.2017.0151. Epub 2017 Sep 20. Mol Cells. 2017. PMID: 28927262 Free PMC article. Review.
-
The multifaceted role of autophagy in cancer and the microenvironment.Med Res Rev. 2019 Mar;39(2):517-560. doi: 10.1002/med.21531. Epub 2018 Oct 9. Med Res Rev. 2019. PMID: 30302772 Free PMC article. Review.
-
Bystander effects of ionizing radiation: conditioned media from X-ray irradiated MCF-7 cells increases the angiogenic ability of endothelial cells.Cell Commun Signal. 2019 Dec 16;17(1):165. doi: 10.1186/s12964-019-0474-8. Cell Commun Signal. 2019. PMID: 31842899 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous
