Proteins of the ciliary axoneme are found on cytoplasmic membrane vesicles during growth of cilia
- PMID: 24814148
- PMCID: PMC4096978
- DOI: 10.1016/j.cub.2014.03.047
Proteins of the ciliary axoneme are found on cytoplasmic membrane vesicles during growth of cilia
Abstract
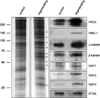
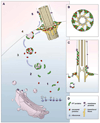
The cilium is a specialized extension of the cell in which many specific proteins are admitted and retained, while many others are excluded or expelled. In order to maintain the organelle, the cell must possess mechanisms for the selective gating of protein entry, as well as for the targeted transport of proteins to the cilium from their sites of synthesis within the cell [1-4]. We hypothesized that the cell employs cytoplasmic vesicles as vehicles not only for the transport of proteins destined for the ciliary membrane but also for the transport of axonemal proteins to the cilium by means of peripheral association with vesicles. To test this hypothesis, we employed two different experimental strategies: (1) isolation and biochemical characterization of cytoplasmic vesicles that carry ciliary proteins, and (2) in situ localization of ciliary proteins on cytoplasmic vesicle surfaces using gold labeling and electron microscopy. Our findings indicate that structural proteins destined for the ciliary axoneme are attached to the outer surfaces of cytoplasmic vesicles that carry integral ciliary membrane proteins during the process of ciliary growth.
Copyright © 2014 Elsevier Ltd. All rights reserved.
Figures


Similar articles
-
Intraflagellar transport: it's not just for cilia anymore.Curr Opin Cell Biol. 2010 Feb;22(1):75-80. doi: 10.1016/j.ceb.2009.10.010. Epub 2009 Dec 3. Curr Opin Cell Biol. 2010. PMID: 19962875 Free PMC article. Review.
-
The ciliary inner dynein arm, I1 dynein, is assembled in the cytoplasm and transported by IFT before axonemal docking.Cytoskeleton (Hoboken). 2014 Oct;71(10):573-86. doi: 10.1002/cm.21192. Epub 2014 Oct 30. Cytoskeleton (Hoboken). 2014. PMID: 25252184 Free PMC article.
-
Transient Internalization and Microtubule-Dependent Trafficking of a Ciliary Signaling Receptor from the Plasma Membrane to the Cilium.Curr Biol. 2019 Sep 9;29(17):2942-2947.e2. doi: 10.1016/j.cub.2019.07.022. Epub 2019 Aug 15. Curr Biol. 2019. PMID: 31422889 Free PMC article.
-
Calcium regulation of ciliary motility analysis of axonemal calcium-binding proteins.Methods Cell Biol. 2009;92:163-80. doi: 10.1016/S0091-679X(08)92011-2. Epub 2009 Nov 21. Methods Cell Biol. 2009. PMID: 20409805
-
Molecular mechanisms of protein and lipid targeting to ciliary membranes.J Cell Sci. 2010 Feb 15;123(Pt 4):529-36. doi: 10.1242/jcs.062968. J Cell Sci. 2010. PMID: 20145001 Free PMC article. Review.
Cited by
-
Two classes of short intraflagellar transport train with different 3D structures are present in Chlamydomonas flagella.J Cell Sci. 2016 May 15;129(10):2064-74. doi: 10.1242/jcs.183244. Epub 2016 Apr 4. J Cell Sci. 2016. PMID: 27044756 Free PMC article.
-
Uni-directional ciliary membrane protein trafficking by a cytoplasmic retrograde IFT motor and ciliary ectosome shedding.Elife. 2015 Feb 17;4:e05242. doi: 10.7554/eLife.05242. Elife. 2015. PMID: 25688564 Free PMC article.
-
Cilia proteins getting to work - how do they commute from the cytoplasm to the base of cilia?J Cell Sci. 2022 Sep 1;135(17):jcs259444. doi: 10.1242/jcs.259444. Epub 2022 Sep 8. J Cell Sci. 2022. PMID: 36073764 Free PMC article. Review.
-
Delivery of intraflagellar transport proteins to the ciliary base and assembly into trains.Sci Adv. 2025 Apr 4;11(14):eadr1716. doi: 10.1126/sciadv.adr1716. Epub 2025 Apr 4. Sci Adv. 2025. PMID: 40184459 Free PMC article.
-
Mechanism of IFT-A polymerization into trains for ciliary transport.Cell. 2022 Dec 22;185(26):4986-4998.e12. doi: 10.1016/j.cell.2022.11.033. Cell. 2022. PMID: 36563665 Free PMC article.
References
-
- Nachury MV, Loktev AV, Zhang Q, Westlake CJ, Peranen J, Merdes A, Slusarski DC, Scheller RH, Bazan JF, Sheffield VC, et al. A core complex of BBS proteins cooperates with the GTPase Rab8 to promote ciliary membrane biogenesis. Cell. 2007;129:1201–1213. - PubMed
-
- Deane JA, Cole DG, Seeley ES, Diener DR, Rosenbaum JL. Localization of intraflagellar transport protein IFT52 identifies basal body transitional fibers as the docking site for IFT particles. Curr. Biol. 2001;11:1586–1590. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

