Matrix metalloproteinase-9-null mice are resistant to TGF-β-induced anterior subcapsular cataract formation
- PMID: 24814605
- PMCID: PMC4076463
- DOI: 10.1016/j.ajpath.2014.03.013
Matrix metalloproteinase-9-null mice are resistant to TGF-β-induced anterior subcapsular cataract formation
Abstract
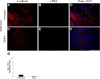
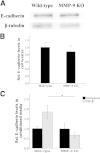
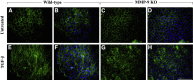
Epithelial-mesenchymal transition (EMT) is associated with fibrotic diseases in the lens, such as anterior subcapsular cataract (ASC) formation. Often mediated by transforming growth factor (TGF)-β, EMT in the lens involves the transformation of lens epithelial cells into a multilayering of myofibroblasts, which manifest as plaques beneath the lens capsule. TGF-β-induced EMT and ASC have been associated with the up-regulation of two matrix metalloproteinases (MMPs): MMP-2 and MMP-9. The current study used MMP-2 and MMP-9 knockout (KO) mice to further determine their unique roles in TGF-β-induced ASC formation. Adenoviral injection of active TGF-β1 into the anterior chamber of all wild-type and MMP-2 KO mice led to the formation of distinct ASC plaques that were positive for α-smooth muscle actin, a marker of EMT. In contrast, only a small proportion of the MMP-9 KO eyes injected with adenovirus-expressing TGF-β1 exhibited ASC plaques. Isolated lens epithelial explants from wild-type and MMP-2 KO mice that were treated with TGF-β exhibited features indicative of EMT, whereas those from MMP-9 KO mice did not acquire a mesenchymal phenotype. MMP-9 KO mice were further bred onto a TGF-β1 transgenic mouse line that exhibits severe ASC formation, but shows a resistance to ASC formation in the absence of MMP-9. These findings suggest that MMP-9 expression is more critical than MMP-2 in mediating TGF-β-induced ASC formation.
Copyright © 2014 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved.
Figures




Similar articles
-
Temporal changes in MMP mRNA expression in the lens epithelium during anterior subcapsular cataract formation.Exp Eye Res. 2009 Feb;88(2):323-30. doi: 10.1016/j.exer.2008.08.014. Epub 2008 Sep 6. Exp Eye Res. 2009. PMID: 18809398 Free PMC article.
-
Lens-specific expression of TGF-beta induces anterior subcapsular cataract formation in the absence of Smad3.Invest Ophthalmol Vis Sci. 2006 Aug;47(8):3450-60. doi: 10.1167/iovs.05-1208. Invest Ophthalmol Vis Sci. 2006. PMID: 16877415 Free PMC article.
-
Arginase-1 promotes lens epithelial-to-mesenchymal transition in different models of anterior subcapsular cataract.Cell Commun Signal. 2023 Sep 18;21(1):236. doi: 10.1186/s12964-023-01210-4. Cell Commun Signal. 2023. PMID: 37723490 Free PMC article.
-
Transforming growth factor-beta-induced epithelial-mesenchymal transition in the lens: a model for cataract formation.Cells Tissues Organs. 2005;179(1-2):43-55. doi: 10.1159/000084508. Cells Tissues Organs. 2005. PMID: 15942192 Review.
-
Regulation of extracellular matrix remodeling following transforming growth factor-beta1/epidermal growth factor-stimulated epithelial-mesenchymal transition in human premalignant keratinocytes.Cells Tissues Organs. 2007;185(1-3):116-22. doi: 10.1159/000101312. Cells Tissues Organs. 2007. PMID: 17587817 Review.
Cited by
-
Immune cells in lens injury repair and fibrosis.Exp Eye Res. 2021 Aug;209:108664. doi: 10.1016/j.exer.2021.108664. Epub 2021 Jun 11. Exp Eye Res. 2021. PMID: 34126081 Free PMC article. Review.
-
The Role of Metalloproteinases and Their Tissue Inhibitors on Ocular Diseases: Focusing on Potential Mechanisms.Int J Mol Sci. 2022 Apr 12;23(8):4256. doi: 10.3390/ijms23084256. Int J Mol Sci. 2022. PMID: 35457074 Free PMC article. Review.
-
Rapamycin relieves the cataract caused by ablation of Gja8b through stimulating autophagy in zebrafish.Autophagy. 2021 Nov;17(11):3323-3337. doi: 10.1080/15548627.2021.1872188. Epub 2021 Jan 20. Autophagy. 2021. PMID: 33472493 Free PMC article.
-
Transforming growth factor-β in tumour development.Front Mol Biosci. 2022 Oct 4;9:991612. doi: 10.3389/fmolb.2022.991612. eCollection 2022. Front Mol Biosci. 2022. PMID: 36267157 Free PMC article. Review.
-
The role of lipocalin-2 in age-related macular degeneration (AMD).Cell Mol Life Sci. 2020 Mar;77(5):835-851. doi: 10.1007/s00018-019-03423-8. Epub 2020 Jan 4. Cell Mol Life Sci. 2020. PMID: 31901947 Free PMC article. Review.
References
-
- World Health Organization . World Health Organization; Geneva, Switzerland: 2000. Blindness: vision 2020: The Global Initiative for the Elimination of Avoidable Blindness. Fact sheet 213.
-
- World Health Organization . World Health Organization; Geneva, Switzerland: 2012. Visual Impairment and Blindness. Fact sheet 282.
-
- Pascolini D., Mariotti S.P. Global estimates of visual impairment: 2010. Br J Ophthalmol. 2012;96:614–618. - PubMed
-
- Allen P.J. Cataract surgery practice and endophthalmitis prevention by Australian and New Zealand ophthalmologists–comment. Clin Exp Ophthalmol. 2007;35:391. - PubMed
-
- West S.K. Looking forward to 20/20: a focus on the epidemiology of eye diseases. Epidemiol Rev. 2000;22:64–70. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

