Downregulation of connexin43 by microRNA-130a in cardiomyocytes results in cardiac arrhythmias
- PMID: 24819345
- PMCID: PMC4412372
- DOI: 10.1016/j.yjmcc.2014.04.024
Downregulation of connexin43 by microRNA-130a in cardiomyocytes results in cardiac arrhythmias
Abstract
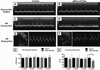
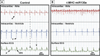
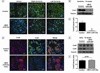
MicroRNAs (miRNAs) are now recognized as critical regulators of diverse physiological and pathological processes; however, studies of miRNAs and arrhythmogenesis remain sparse. Connexin43 (Cx43), a major cardiac gap junction protein, has elicited great interest in its role in arrhythmias. Additionally, Cx43 was a potential target for miR-130a as predicted by several computational algorithms. This study investigates the effect of miR-130a overexpression in the adult heart and its effect on cardiac rhythm. Using a cardiac-specific inducible system, transgenic mice demonstrated both atrial and ventricular arrhythmias. We performed ventricular-programmed electrical stimulation and found that the αMHC-miR130a mice developed sustained ventricular tachycardia beginning 6weeks after overexpression. Western blot analysis demonstrated a steady decline in Cx43 after 2weeks of overexpression with over a 90% reduction in Cx43 levels by 10weeks. Immunofluorescent staining confirmed a near complete loss of Cx43 throughout the heart. To validate Cx43 as a direct target of miR-130a, we performed in vitro target assays in 3T3 fibroblasts and HL-1 cardiomyocytes, both known to endogenously express miR-130a. Using a luciferase reporter fused to the 3'UTR of Cx43, we found a 52.9% reduction in luciferase activity in 3T3 cells (p<0.0001) and a 47.6% reduction in HL-1 cells (p=0.0056) compared to controls. Addition of an antisense miR-130a inhibitor resulted in a loss of inhibitory activity of the Cx43 3'UTR reporter. We have identified an unappreciated role for miR-130a as a direct regulator of Cx43. Overexpression of miR-130a may contribute importantly to gap junction remodeling and to the pathogenesis of atrial and ventricular arrhythmias.
Keywords: Atrial arrhythmia; Connexin43; MicroRNA; Ventricular arrhythmia.
Copyright © 2014 Elsevier Ltd. All rights reserved.
Conflict of interest statement
No competing potential conflicts exist.
Figures



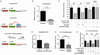
Similar articles
-
MicroRNA-206 Downregulates Connexin43 in Cardiomyocytes to Induce Cardiac Arrhythmias in a Transgenic Mouse Model.Heart Lung Circ. 2019 Nov;28(11):1755-1761. doi: 10.1016/j.hlc.2018.09.008. Epub 2018 Oct 4. Heart Lung Circ. 2019. PMID: 30322759
-
MicroRNA-130a Regulation of Desmocollin 2 in a Novel Model of Arrhythmogenic Cardiomyopathy.Microrna. 2017;6(2):143-150. doi: 10.2174/2211536605666161109111031. Microrna. 2017. PMID: 27834139 Free PMC article.
-
Cardiomyocyte-specific overexpression of the ubiquitin ligase Wwp1 contributes to reduction in Connexin 43 and arrhythmogenesis.J Mol Cell Cardiol. 2015 Nov;88:1-13. doi: 10.1016/j.yjmcc.2015.09.004. Epub 2015 Sep 16. J Mol Cell Cardiol. 2015. PMID: 26386426 Free PMC article.
-
Intracellular trafficking pathways of Cx43 gap junction channels.Biochim Biophys Acta Biomembr. 2018 Jan;1860(1):40-47. doi: 10.1016/j.bbamem.2017.05.018. Epub 2017 May 30. Biochim Biophys Acta Biomembr. 2018. PMID: 28576298 Free PMC article. Review.
-
[Remodeling of cardiac gap junctions and arrhythmias].Sheng Li Xue Bao. 2011 Dec 25;63(6):586-92. Sheng Li Xue Bao. 2011. PMID: 22193455 Review. Chinese.
Cited by
-
Knockdown of the long non‑coding RNA MALAT1 ameliorates TNF‑α‑mediated endothelial cell pyroptosis via the miR‑30c‑5p/Cx43 axis.Mol Med Rep. 2023 Apr;27(4):90. doi: 10.3892/mmr.2023.12977. Epub 2023 Mar 10. Mol Med Rep. 2023. PMID: 36896775 Free PMC article.
-
Role of MicroRNAs in Diagnosis, Prognosis, and Treatment of Acute Heart Failure: Ambassadors from Intracellular Zone.Galen Med J. 2020 Jun 23;9:e1818. doi: 10.31661/gmj.v9i0.1818. eCollection 2020. Galen Med J. 2020. PMID: 34466598 Free PMC article. Review.
-
Mechanism of Nardostachyos Radix et Rhizoma-Salidroside in the treatment of premature ventricular beats based on network pharmacology and molecular docking.Sci Rep. 2023 Nov 25;13(1):20741. doi: 10.1038/s41598-023-48277-0. Sci Rep. 2023. PMID: 38007574 Free PMC article.
-
MicroRNA-130a Increases and Predicts Cardiotoxicity during Adjuvant Chemotherapy in Human Epidermal Growth Factor Receptor-2-Positive Breast Cancer.J Breast Cancer. 2021 Apr;24(2):153-163. doi: 10.4048/jbc.2021.24.e15. Epub 2021 Mar 2. J Breast Cancer. 2021. PMID: 33818020 Free PMC article.
-
Decreased expression of microRNA-130a correlates with TNF-α in the development of osteoarthritis.Int J Clin Exp Pathol. 2015 Mar 1;8(3):2555-64. eCollection 2015. Int J Clin Exp Pathol. 2015. PMID: 26045761 Free PMC article.
References
-
- Nattel S, Maguy A, Le Bouter S, Yeh YH. Arrhythmogenic ion-channel remodeling in the heart: heart failure, myocardial infarction, and atrial fibrillation. Physiol Rev. 2007;87:425–456. - PubMed
-
- Peters NS, Coromilas J, Severs NJ, Wit AL. Disturbed connexin43 gap junction distribution correlates with the location of reentrant circuits in the epicardial border zone of healing canine infarcts that cause ventricular tachycardia. Circulation. 1997;95:988–996. - PubMed
-
- Yao JA, Hussain W, Patel P, Peters NS, Boyden PA, Wit AL. Remodeling of gap junctional channel function in epicardial border zone of healing canine infarcts. Circ Res. 2003;92:437–443. - PubMed
-
- Willecke K, Eiberger J, Degen J, Eckardt D, Romualdi A, Guldenagel M, et al. Structural and functional diversity of connexin genes in the mouse and human genome. Biol Chem. 2002;383:725–737. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

