Phase III trials of new oral anticoagulants in the acute treatment and secondary prevention of VTE: comparison and critique of study methodology and results
- PMID: 24821599
- PMCID: PMC4033807
- DOI: 10.1007/s12325-014-0119-7
Phase III trials of new oral anticoagulants in the acute treatment and secondary prevention of VTE: comparison and critique of study methodology and results
Abstract
The traditional treatment of venous thromboembolism (VTE) has been use of heparin and vitamin K antagonists (VKA), and although shown to be effective, they have numerous limitations. New oral anticoagulants (NOACs) including direct thrombin (factor IIa) inhibitors (dabigatran) and selective factor Xa inhibitors (rivaroxaban, apixaban and edoxaban) have emerged as promising alternatives with the potential to overcome the limitations of traditional treatments. Clinical trials have been performed with a view to making significant changes to the acute, long-term and extended treatment of VTE. Data are now available on the efficacy and safety, including bleeding rates, of the NOACs in comparison with VKA in the acute treatment and secondary prevention of VTE as well as in comparison with placebo extended VTE treatment. This review compares and contrasts the design and results of the Phase III trials of NOACs in VTE and discusses the implications of the NOACs in terms of treatment strategies in VTE patients.
Figures
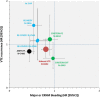
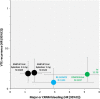
References
-
- Cohen AT, Agnelli G, Anderson FA, et al. Venous thromboembolism (VTE) in Europe. The number of VTE events and associated morbidity and mortality. Thromb Haemost. 2007;98:756–764. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

