Repression of cyclin D1 expression is necessary for the maintenance of cell cycle exit in adult mammalian cardiomyocytes
- PMID: 24821722
- PMCID: PMC4140299
- DOI: 10.1074/jbc.M113.541953
Repression of cyclin D1 expression is necessary for the maintenance of cell cycle exit in adult mammalian cardiomyocytes
Abstract
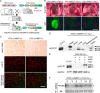
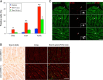
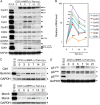
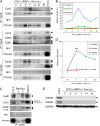
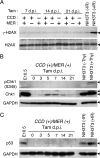
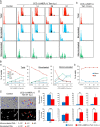
The hearts of neonatal mice and adult zebrafish can regenerate after injury through proliferation of preexisting cardiomyocytes. However, adult mammals are not capable of cardiac regeneration because almost all cardiomyocytes exit their cell cycle. Exactly how the cell cycle exit is maintained and how many adult cardiomyocytes have the potential to reenter the cell cycle are unknown. The expression and activation levels of main cyclin-cyclin-dependent kinase (CDK) complexes are extremely low or undetectable at adult stages. The nuclear DNA content of almost all cardiomyocytes is 2C, indicating the cell cycle exit from G1-phase. Here, we induced expression of cyclin D1, which regulates the progression of G1-phase, only in differentiated cardiomyocytes of adult mice. In these cardiomyocytes, S-phase marker-positive cardiomyocytes and the expression of main cyclins and CDKs increased remarkably, although cyclin B1-CDK1 activation was inhibited in an ATM/ATR-independent manner. The phosphorylation pattern of CDK1 and expression pattern of Cdc25 subtypes suggested that a deficiency in the increase in Cdc25 (a and -b), which is required for M-phase entry, inhibited the cyclin B1-CDK1 activation. Finally, analysis of cell cycle distribution patterns showed that >40% of adult mouse cardiomyocytes reentered the cell cycle by the induction of cyclin D1. The cell cycle of these binucleated cardiomyocytes was arrested before M-phase, and many mononucleated cardiomyocytes entered endoreplication. These data indicate that silencing the cyclin D1 expression is necessary for the maintenance of the cell cycle exit and suggest a mechanism that involves inhibition of M-phase entry.
Keywords: Cardiac Muscle; Cell Cycle; Cyclin D1; Gene Silencing; Regeneration.
© 2014 by The American Society for Biochemistry and Molecular Biology, Inc.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

