Water sensor ppk28 modulates Drosophila lifespan and physiology through AKH signaling
- PMID: 24821805
- PMCID: PMC4050545
- DOI: 10.1073/pnas.1315461111
Water sensor ppk28 modulates Drosophila lifespan and physiology through AKH signaling
Abstract
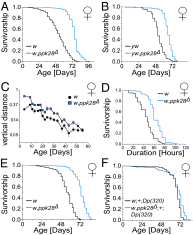
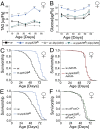
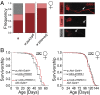
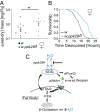
Sensory perception modulates lifespan across taxa, presumably due to alterations in physiological homeostasis after central nervous system integration. The coordinating circuitry of this control, however, remains unknown. Here, we used the Drosophila melanogaster gustatory system to dissect one component of sensory regulation of aging. We found that loss of the critical water sensor, pickpocket 28 (ppk28), altered metabolic homeostasis to promote internal lipid and water stores and extended healthy lifespan. Additionally, loss of ppk28 increased neuronal glucagon-like adipokinetic hormone (AKH) signaling, and the AKH receptor was necessary for ppk28 mutant effects. Furthermore, activation of AKH-producing cells alone was sufficient to enhance longevity, suggesting that a perceived lack of water availability triggers a metabolic shift that promotes the production of metabolic water and increases lifespan via AKH signaling. This work provides an example of how discrete gustatory signals recruit nutrient-dependent endocrine systems to coordinate metabolic homeostasis, thereby influencing long-term health and aging.
Keywords: adipokinetic hormone signaling; taste.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Regulation of insulin and adipokinetic hormone/glucagon production in flies.Wiley Interdiscip Rev Dev Biol. 2020 Mar;9(2):e360. doi: 10.1002/wdev.360. Epub 2019 Aug 4. Wiley Interdiscip Rev Dev Biol. 2020. PMID: 31379062 Free PMC article. Review.
-
Impact of high-fat diet on lifespan, metabolism, fecundity and behavioral senescence in Drosophila.Insect Biochem Mol Biol. 2021 Jun;133:103495. doi: 10.1016/j.ibmb.2020.103495. Epub 2020 Nov 7. Insect Biochem Mol Biol. 2021. PMID: 33171202
-
Energy Homeostasis Control in Drosophila Adipokinetic Hormone Mutants.Genetics. 2015 Oct;201(2):665-83. doi: 10.1534/genetics.115.178897. Epub 2015 Aug 14. Genetics. 2015. PMID: 26275422 Free PMC article.
-
Role of adipokinetic hormone during starvation in Drosophila.Comp Biochem Physiol B Biochem Mol Biol. 2018 Dec;226:26-35. doi: 10.1016/j.cbpb.2018.08.004. Epub 2018 Aug 12. Comp Biochem Physiol B Biochem Mol Biol. 2018. PMID: 30110658
-
The Intrinsic Nutrient Sensing Adipokinetic Hormone Producing Cells Function in Modulation of Metabolism, Activity, and Stress.Int J Mol Sci. 2021 Jul 13;22(14):7515. doi: 10.3390/ijms22147515. Int J Mol Sci. 2021. PMID: 34299134 Free PMC article. Review.
Cited by
-
Sensory perception of dead conspecifics induces aversive cues and modulates lifespan through serotonin in Drosophila.Nat Commun. 2019 May 30;10(1):2365. doi: 10.1038/s41467-019-10285-y. Nat Commun. 2019. PMID: 31147540 Free PMC article.
-
Slowed aging during reproductive dormancy is reflected in genome-wide transcriptome changes in Drosophila melanogaster.BMC Genomics. 2016 Jan 13;17:50. doi: 10.1186/s12864-016-2383-1. BMC Genomics. 2016. PMID: 26758761 Free PMC article.
-
The sugar-responsive enteroendocrine neuropeptide F regulates lipid metabolism through glucagon-like and insulin-like hormones in Drosophila melanogaster.Nat Commun. 2021 Aug 10;12(1):4818. doi: 10.1038/s41467-021-25146-w. Nat Commun. 2021. PMID: 34376687 Free PMC article.
-
AKH Signaling in D. melanogaster Alters Larval Development in a Nutrient-Dependent Manner That Influences Adult Metabolism.Front Physiol. 2021 Feb 23;12:619219. doi: 10.3389/fphys.2021.619219. eCollection 2021. Front Physiol. 2021. PMID: 33708137 Free PMC article.
-
Adaptive Physiological Response to Perceived Scarcity as a Mechanism of Sensory Modulation of Life Span.J Gerontol A Biol Sci Med Sci. 2015 Sep;70(9):1088-91. doi: 10.1093/gerona/glv039. Epub 2015 Apr 15. J Gerontol A Biol Sci Med Sci. 2015. PMID: 25878032 Free PMC article.
References
-
- Alcedo J, Kenyon C. Regulation of C. elegans longevity by specific gustatory and olfactory neurons. Neuron. 2004;41(1):45–55. - PubMed
-
- Libert S, Pletcher SD. Modulation of longevity by environmental sensing. Cell. 2007;131(7):1231–1234. - PubMed
-
- Apfeld J, Kenyon C. Regulation of lifespan by sensory perception in Caenorhabditis elegans. Nature. 1999;402(6763):804–809. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- T-32-AG000114/AG/NIA NIH HHS/United States
- T32 AG000114/AG/NIA NIH HHS/United States
- R-01-AG023166/AG/NIA NIH HHS/United States
- 5-T32-GM007863-32/GM/NIGMS NIH HHS/United States
- R01 AG030593/AG/NIA NIH HHS/United States
- R-01-AG030593/AG/NIA NIH HHS/United States
- T32 GM007863/GM/NIGMS NIH HHS/United States
- R01 AG043972/AG/NIA NIH HHS/United States
- F-32-AG042253/AG/NIA NIH HHS/United States
- P30 DK020572/DK/NIDDK NIH HHS/United States
- P30-AG-013283/AG/NIA NIH HHS/United States
- T32 GM007315/GM/NIGMS NIH HHS/United States
- F32 AG042253/AG/NIA NIH HHS/United States
- R-01-AG043972/AG/NIA NIH HHS/United States
- R01 AG023166/AG/NIA NIH HHS/United States
- P30 AG013283/AG/NIA NIH HHS/United States
- 5-T-32-GM007315/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

