Trop-2 is a determinant of breast cancer survival
- PMID: 24824621
- PMCID: PMC4019539
- DOI: 10.1371/journal.pone.0096993
Trop-2 is a determinant of breast cancer survival
Erratum in
- PLoS One. 2014;9(10):e110606
Abstract
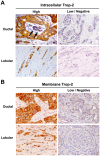
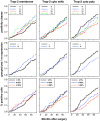
Trop-2 is a calcium signal transducer that drives tumor growth. Anti-Trop-2 antibodies with selective reactivity versus Trop-2 maturation stages allowed to identify two different pools of Trop-2, one localized in the cell membrane and one in the cytoplasm. Of note, membrane-localized/functional Trop-2 was found to be differentially associated with determinants of tumor aggressiveness and distinct breast cancer subgroups. These findings candidated Trop-2 states to having an impact on cancer progression. We tested this model in breast cancer. A large, consecutive human breast cancer case series (702 cases; 8 years median follow-up) was analyzed by immunohistochemistry with anti-Trop-2 antibodies with selective reactivity for cytoplasmic-retained versus functional, membrane-associated Trop-2. We show that membrane localization of Trop-2 is an unfavorable prognostic factor for overall survival (1+ versus 0 for all deaths: hazard ratio, 1.63; P = 0.04), whereas intracellular Trop-2 has a favorable impact on prognosis, with an adjusted hazard ratio for all deaths of 0.48 (high versus low; P = 0.003). A corresponding impact of intracellular Trop-2 was found on disease relapse (high versus low: hazard ratio, 0.51; P = 0.004). Altogether, we demonstrate that the Trop-2 activation states are critical determinants of tumor progression and are powerful indicators of breast cancer patients survival.
Conflict of interest statement
Figures



References
-
- Ambrogi F, Biganzoli E, Querzoli P, Ferretti S, Boracchi P, et al. (2006) Molecular subtyping of breast cancer from traditional tumor marker profiles using parallel clustering methods. Clin Cancer Res 12: 781–790. - PubMed
-
- Cimoli G, Malacarne D, Ponassi R, Valenti M, Alberti S, et al. (2004) Meta-analysis of the role of p53 status in isogenic systems tested for sensitivity to cytotoxic antineoplastic drugs. Biochim Biophys Acta 1705: 103–120. - PubMed
-
- Biganzoli E, Pedriali M, Querzoli P, Nenci I, Iacobelli S, et al. (2010) Sentinel Node and Bone Marrow Micrometastases and Nanometastases. Curr Breast Cancer Rep 2: 96–106.
-
- Tinari N, Lattanzio R, Natoli C, Cianchetti E, Angelucci D, et al. (2006) Changes of topoisomerase IIalpha expression in breast tumors after neoadjuvant chemotherapy predicts relapse-free survival. Clin Cancer Res 12: 1501–1506. - PubMed
-
- Fornaro M, Dell'Arciprete R, Stella M, Bucci C, Nutini M, et al. (1995) Cloning of the gene encoding TROP-2, a cell-surface glycoprotein expressed by human carcinomas. Int J Cancer 62: 610–618. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

