Pannexins form gap junctions with electrophysiological and pharmacological properties distinct from connexins
- PMID: 24828343
- PMCID: PMC4021813
- DOI: 10.1038/srep04955
Pannexins form gap junctions with electrophysiological and pharmacological properties distinct from connexins
Abstract
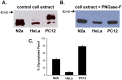
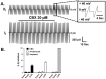
Stable expression of pannexin 1 (Panx1) and pannexin 3 (Panx3) resulted in functional gap junctions (GJs) in HeLa cells, but not in Neuro-2a (N2a) or PC-12 cells. The glycosylation pattern of expressed Panx1 varied greatly among different cell lines. In contrast to connexin (Cx) containing GJs (Cx-GJs), junctional conductance (Gj) of pannexin GJs (Panx-GJs) is very less sensitive to junctional voltage. Both Panx1 and Panx3 junctions favoured anionic dyes over cations to permeate. Though, carbenoxolone (CBX) and probenecid blocked Panx1 hemichannel activity, they had no effect on Panx1-GJs or Panx3-GJs. Extracellular loop 1 (E1) of Panx1 possibly bears the binding pocket. The Cx-GJ blocker heptanol blocked neither Panx1 hemichannel nor Panx-GJs. Unlike the GJs formed by most Cxs, CO2 did not uncouple Panx-GJs completely. Oxygen and glucose deprivation (OGD) caused lesser uncoupling of Panx-GJs compared to Cx43-GJs. These findings demonstrate properties of Panx-GJs that are distinctly different from Cx-GJs.
Figures



Similar articles
-
Pannexin 1 and pannexin 3 are glycoproteins that exhibit many distinct characteristics from the connexin family of gap junction proteins.J Cell Sci. 2007 Nov 1;120(Pt 21):3772-83. doi: 10.1242/jcs.009514. Epub 2007 Oct 9. J Cell Sci. 2007. PMID: 17925379
-
Functional analysis of hemichannels and gap-junctional channels formed by connexins 43 and 46.Mol Vis. 2010 Jul 15;16:1343-52. Mol Vis. 2010. PMID: 20664797 Free PMC article.
-
Junctional delay, frequency, and direction-dependent uncoupling of human heterotypic Cx45/Cx43 gap junction channels.J Mol Cell Cardiol. 2017 Oct;111:17-26. doi: 10.1016/j.yjmcc.2017.07.117. Epub 2017 Jul 29. J Mol Cell Cardiol. 2017. PMID: 28760564
-
Mechanisms of Pannexin 1 (PANX1) Channel Mechanosensitivity and Its Pathological Roles.Int J Mol Sci. 2022 Jan 28;23(3):1523. doi: 10.3390/ijms23031523. Int J Mol Sci. 2022. PMID: 35163442 Free PMC article. Review.
-
Roles of gap junctions, connexins, and pannexins in epilepsy.Front Physiol. 2014 May 7;5:172. doi: 10.3389/fphys.2014.00172. eCollection 2014. Front Physiol. 2014. PMID: 24847276 Free PMC article. Review.
Cited by
-
Astroglial Connexins in Neurodegenerative Diseases.Front Mol Neurosci. 2021 May 28;14:657514. doi: 10.3389/fnmol.2021.657514. eCollection 2021. Front Mol Neurosci. 2021. PMID: 34122008 Free PMC article. Review.
-
Pannexin 1 Transgenic Mice: Human Diseases and Sleep-Wake Function Revision.Int J Mol Sci. 2021 May 17;22(10):5269. doi: 10.3390/ijms22105269. Int J Mol Sci. 2021. PMID: 34067798 Free PMC article.
-
Single channel properties of pannexin-1 and connexin-43 hemichannels and P2X7 receptors in astrocytes cultured from rodent spinal cords.Glia. 2022 Dec;70(12):2260-2275. doi: 10.1002/glia.24250. Epub 2022 Aug 2. Glia. 2022. PMID: 35915989 Free PMC article.
-
Enhanced Methodologies for Investigating the Electrophysiological Characteristics of Endogenous Pannexin 1 Intercellular Cell-Cell Channels.Methods Mol Biol. 2024;2801:135-145. doi: 10.1007/978-1-0716-3842-2_11. Methods Mol Biol. 2024. PMID: 38578419
-
Role of Astroglial Hemichannels and Pannexons in Memory and Neurodegenerative Diseases.Front Integr Neurosci. 2016 Jul 20;10:26. doi: 10.3389/fnint.2016.00026. eCollection 2016. Front Integr Neurosci. 2016. PMID: 27489539 Free PMC article. Review.
References
-
- Panchin Y. et al. A ubiquitous family of putative gap junction molecules. Curr Biol 10, R473–474 (2000). - PubMed
-
- Baranova A. et al. The mammalian pannexin family is homologous to the invertebrate innexin gap junction proteins. Genomics 83, 706–716 (2004). - PubMed
-
- Panchin Y. V. Evolution of gap junction proteins--the pannexin alternative. J Exp Biol 208, 1415–1419 (2005). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

