Human lung epithelial cells contain Mycobacterium tuberculosis in a late endosomal vacuole and are efficiently recognized by CD8⁺ T cells
- PMID: 24828674
- PMCID: PMC4020835
- DOI: 10.1371/journal.pone.0097515
Human lung epithelial cells contain Mycobacterium tuberculosis in a late endosomal vacuole and are efficiently recognized by CD8⁺ T cells
Abstract
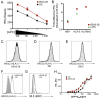
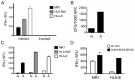
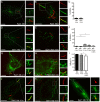
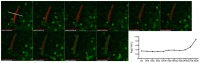
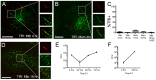
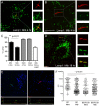
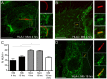
Mycobacterium tuberculosis (Mtb) is transmitted via inhalation of aerosolized particles. While alveolar macrophages are thought to play a central role in the acquisition and control of this infection, Mtb also has ample opportunity to interact with the airway epithelium. In this regard, we have recently shown that the upper airways are enriched with a population of non-classical, MR1-restricted, Mtb-reactive CD8⁺ T cells (MAIT cells). Additionally, we have demonstrated that Mtb-infected epithelial cells lining the upper airways are capable of stimulating IFNγ production by MAIT cells. In this study, we demonstrate that airway epithelial cells efficiently stimulate IFNγ release by MAIT cells as well as HLA-B45 and HLA-E restricted T cell clones. Characterization of the intracellular localization of Mtb in epithelial cells indicates that the vacuole occupied by Mtb in epithelial cells is distinct from DC in that it acquires Rab7 molecules and does not retain markers of early endosomes such as Rab5. The Mtb vacuole is also heterogeneous as there is a varying degree of association with Lamp1 and HLA-I. Although the Mtb vacuole shares markers associated with the late endosome, it does not acidify, and the bacteria are able to replicate within the cell. This work demonstrates that Mtb infected lung epithelial cells are surprisingly efficient at stimulating IFNγ release by CD8⁺ T cells.
Conflict of interest statement
Figures
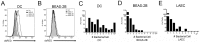
References
-
- Corbett EL, Watt CJ, Walker N, Maher D, Williams BG, et al. (2003) The growing burden of tuberculosis: global trends and interactions with the HIV epidemic. Arch Intern Med 163: 1009–1021. - PubMed
-
- Reichman LB, Hershfield ES (2000) Lung biology in health and disease. Tuberculosis: a comprehensive international approach. 2nd ed. New York: Dekker. pp. 898.
-
- Verver S, Warren RM, Munch Z, Richardson M, van der Spuy GD, et al. (2004) Proportion of tuberculosis transmission that takes place in households in a high-incidence area. Lancet 363: 212–214. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

