The Hippo pathway controls a switch between retinal progenitor cell proliferation and photoreceptor cell differentiation in zebrafish
- PMID: 24828882
- PMCID: PMC4020862
- DOI: 10.1371/journal.pone.0097365
The Hippo pathway controls a switch between retinal progenitor cell proliferation and photoreceptor cell differentiation in zebrafish
Abstract
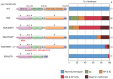
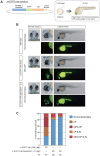
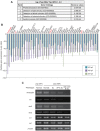
The precise regulation of numbers and types of neurons through control of cell cycle exit and terminal differentiation is an essential aspect of neurogenesis. The Hippo signaling pathway has recently been identified as playing a crucial role in promoting cell cycle exit and terminal differentiation in multiple types of stem cells, including in retinal progenitor cells. When Hippo signaling is activated, the core Mst1/2 kinases activate the Lats1/2 kinases, which in turn phosphorylate and inhibit the transcriptional cofactor Yap. During mouse retinogenesis, overexpression of Yap prolongs progenitor cell proliferation, whereas inhibition of Yap decreases this proliferation and promotes retinal cell differentiation. However, to date, it remains unknown how the Hippo pathway affects the differentiation of distinct neuronal cell types such as photoreceptor cells. In this study, we investigated whether Hippo signaling regulates retinogenesis during early zebrafish development. Knockdown of zebrafish mst2 induced early embryonic defects, including altered retinal pigmentation and morphogenesis. Similar abnormal retinal phenotypes were observed in zebrafish embryos injected with a constitutively active form of yap [(yap (5SA)]. Loss of Yap's TEAD-binding domain, two WW domains, or transcription activation domain attenuated the retinal abnormalities induced by yap (5SA), indicating that all of these domains contribute to normal retinal development. Remarkably, yap (5SA)-expressing zebrafish embryos displayed decreased expression of transcription factors such as otx5 and crx, which orchestrate photoreceptor cell differentiation by activating the expression of rhodopsin and other photoreceptor cell genes. Co-immunoprecipitation experiments revealed that Rx1 is a novel interacting partner of Yap that regulates photoreceptor cell differentiation. Our results suggest that Yap suppresses the differentiation of photoreceptor cells from retinal progenitor cells by repressing Rx1-mediated transactivation of photoreceptor cell genes during zebrafish retinogenesis.
Conflict of interest statement
Figures




References
-
- Livesey FJ, Cepko CL (2001) Vertebrate neural cell-fate determination: lessons from the retina. Nat Rev Neurosci 2: 109–118. - PubMed
-
- Dowling JE (2012) The retina : an approachable part of the brain. Cambridge, Mass.: Belknap Press of Harvard University Press.: xvi, 355 p. p.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

