Inhibition of phosphatase and tensin homologue (PTEN) in human ovary in vitro results in increased activation of primordial follicles but compromises development of growing follicles
- PMID: 24830779
- PMCID: PMC4106636
- DOI: 10.1093/molehr/gau037
Inhibition of phosphatase and tensin homologue (PTEN) in human ovary in vitro results in increased activation of primordial follicles but compromises development of growing follicles
Abstract
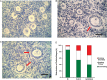
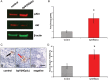
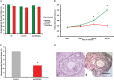
In the mammalian ovary a small number of follicles are steadily recruited from the quiescent pool to undergo development. Follicle loss, maintenance and growth are strictly controlled by complex molecular interactions including the phosphoinositide 3-kinase (PI3K)-protein kinase B (Akt) signalling pathway. Stimulation of PI3K promotes phosphorylation of Akt resulting in follicle survival and activation of growth whereas this pathway is suppressed by the actions of the phosphatase and tensin homologue (PTEN). The aim of this study was to determine the effect of dipotassium bisperoxo(5-hydroxypyridine-2-carboxyl)oxovanadate (bpV), a reversible inhibitor of PTEN, on the activation, survival and development of human ovarian follicles in vitro. Biopsied ovarian tissue fragments were obtained from 17 women aged 23-46 years and exposed to 1 µM bpV(HOpic) (n = 146) or control medium (n = 128) for 24 h. Media were then replaced with control medium and all tissue incubated for a further 5 days. Ovarian tissue from each treatment group was fixed after the initial 24 h culture period and phosphorylated Akt was quantified by western blotting. After 6 days incubation all tissue fragments were inspected under light microscopy and any secondary follicles ≥100 µm isolated. Isolated follicles were cultured individually in control medium supplemented with 100 ng/ml recombinant human activin A. Tissue fragments without follicles suitable for isolation were fixed and processed for histological and immunohistochemical analysis. During 6 days culture, follicle activation occurred in tissue samples from both treatment groups but with significantly more follicles progressing to the secondary stage of development in the presence of 1 µM bpV(HOpic) compared with control (31 versus 16%; P < 0.05). Increased activation was associated with increased Akt phosphorylation and increased nuclear export of FOXO3. However isolated and cultured follicles that had been exposed to bpV(HOpic) showed limited growth and reduced survival compared with follicles from control fragments (P < 0.05). This study demonstrates that inhibition of PTEN with bpV(HOpic) affects human ovarian follicle development by promoting the initiation of follicle growth and development to the secondary stage, as in rodent species, but severely compromises the survival of isolated secondary follicles.
Keywords: PTEN; follicle; human; in vitro; ovary.
© The Author 2014. Published by Oxford University Press on behalf of the European Society of Human Reproduction and Embryology.
Figures
Similar articles
-
Crosstalk between PTEN/PI3K/Akt Signalling and DNA Damage in the Oocyte: Implications for Primordial Follicle Activation, Oocyte Quality and Ageing.Cells. 2020 Jan 14;9(1):200. doi: 10.3390/cells9010200. Cells. 2020. PMID: 31947601 Free PMC article. Review.
-
Inhibition of PTEN activates bovine non-growing follicles in vitro but increases DNA damage and reduces DNA repair response.Hum Reprod. 2019 Feb 1;34(2):297-307. doi: 10.1093/humrep/dey354. Hum Reprod. 2019. PMID: 30521029 Free PMC article.
-
The effect of bpV(HOpic) on in vitro activation of primordial follicles in cultured swine ovarian cortical strips.Reprod Domest Anim. 2019 Aug;54(8):1057-1063. doi: 10.1111/rda.13466. Epub 2019 Jun 14. Reprod Domest Anim. 2019. PMID: 31087697
-
Dynamics of PI3K and Hippo signaling pathways during in vitro human follicle activation.Hum Reprod. 2018 Sep 1;33(9):1705-1714. doi: 10.1093/humrep/dey250. Hum Reprod. 2018. PMID: 30032281
-
Current Understandings of Core Pathways for the Activation of Mammalian Primordial Follicles.Cells. 2021 Jun 13;10(6):1491. doi: 10.3390/cells10061491. Cells. 2021. PMID: 34199299 Free PMC article. Review.
Cited by
-
Stem-Cell-Derived Extracellular Vesicles: Unlocking New Possibilities for Treating Diminished Ovarian Reserve and Premature Ovarian Insufficiency.Life (Basel). 2023 Nov 23;13(12):2247. doi: 10.3390/life13122247. Life (Basel). 2023. PMID: 38137848 Free PMC article. Review.
-
Follicular growth after xenotransplantation of cryopreserved/thawed human ovarian tissue in SCID mice: dynamics and molecular aspects.J Assist Reprod Genet. 2016 Dec;33(12):1585-1593. doi: 10.1007/s10815-016-0769-2. Epub 2016 Jul 27. J Assist Reprod Genet. 2016. PMID: 27465301 Free PMC article.
-
Untapped Reserves: Controlling Primordial Follicle Growth Activation.Trends Mol Med. 2018 Mar;24(3):319-331. doi: 10.1016/j.molmed.2018.01.008. Epub 2018 Feb 13. Trends Mol Med. 2018. PMID: 29452791 Free PMC article. Review.
-
Crosstalk between PTEN/PI3K/Akt Signalling and DNA Damage in the Oocyte: Implications for Primordial Follicle Activation, Oocyte Quality and Ageing.Cells. 2020 Jan 14;9(1):200. doi: 10.3390/cells9010200. Cells. 2020. PMID: 31947601 Free PMC article. Review.
-
An Update on Physiopathological Roles of Akt in the ReprodAKTive Mammalian Ovary.Life (Basel). 2024 Jun 2;14(6):722. doi: 10.3390/life14060722. Life (Basel). 2024. PMID: 38929705 Free PMC article. Review.
References
-
- Abir R, Franks S, Mobberley MA, Moore PA, Margara RA, Winston RM. Mechanical isolation and in vitro growth of preantral and small antral human follicles. Fertil Steril. 1997;68:682–688. - PubMed
-
- Abir R, Roizman P, Fisch B, Nitke S, Okon E, Orvieto R, Ben Rafael Z. study of isolated early human follicles cultured in collagen gels for 24 hours. Hum Reprod. 1999;14:1299–1301. - PubMed
-
- Alak BM, Coskun S, Friedman CI, Kennard EA, Kim MH, Seifer DB. Activin A stimulates meiotic maturation of human oocytes and modulates granulosa cell steroidogenesis in vitro. Fertil Steril. 1998;70:1126–1130. - PubMed
-
- Albertini DF, Combelles CM, Benecchi E, Carabatsos MJ. Cellular basis for paracrine regulation of ovarian follicle development. Reproduction. 2001;121:647–653. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

