Circulating tumor cells predict survival in early average-to-high risk breast cancer patients
- PMID: 24832787
- PMCID: PMC4112925
- DOI: 10.1093/jnci/dju066
Circulating tumor cells predict survival in early average-to-high risk breast cancer patients
Erratum in
- J Natl Cancer Inst. 2014 Sep;106(9):doi/10.1093/jnci/dju273
Abstract
Background: Circulating tumor cells (CTCs) have been shown to predict reduced survival outcomes in metastatic breast cancer.
Methods: CTCs were analyzed in 2026 patients with early breast cancer before adjuvant chemotherapy and in 1492 patients after chemotherapy using the CellSearch System. After immuno-magnetic enrichment for cells expressing the epithelial-cell adhesion molecule, CTCs were defined as nucleated cells expressing cytokeratin and lacking CD45. The patients were followed for a median of 35 months (range = 0-54). Kaplan-Meier analyses and the log-rank test were used for survival analyses. All statistical tests were two-sided.
Results: Before chemotherapy, CTCs were detected in 21.5% of patients (n = 435 of 2026), with 19.6% (n = 136 of 692) of node-negative and 22.4% (n = 299 of 1334) of node-positive patients showing CTCs (P < .001). No association was found with tumor size, grading, or hormone receptor status. After chemotherapy, 22.1% of patients (n = 330 of 1493) were CTC positive. The presence of CTCs was associated with poor disease-free survival (DFS; P < .0001), distant DFS (P < .001), breast cancer-specific survival (P = .008), and overall survival (OS; P = .0002). CTCs were confirmed as independent prognostic markers in multivariable analysis for DFS (hazard ratio [HR] = 2.11; 95% confidence interval [CI] = 1.49 to 2.99; P < .0001) and OS (HR = 2.18; 95% CI = 1.32 to 3.59; P = .002). The prognosis was worst in patients with at least five CTCs per 30 mL blood (DFS: HR = 4.51, 95% CI = 2.59 to 7.86; OS: HR = 3.60, 95% CI = 1.56 to 8.45). The presence of persisting CTCs after chemotherapy showed a negative influence on DFS (HR = 1.12; 95% CI = 1.02 to 1.25; P = .02) and on OS (HR = 1.16; 95% CI = 0.99 to 1.37; P = .06) CONCLUSIONS: These results suggest the independent prognostic relevance of CTCs both before and after adjuvant chemotherapy in a large prospective trial of patients with primary breast cancer.
© The Author 2014. Published by Oxford University Press.
Figures
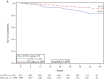



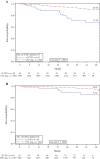
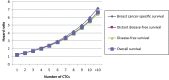

Comment in
-
Circulating tumor cells: what goes around, comes around.J Natl Cancer Inst. 2014 May 15;106(5):dju108. doi: 10.1093/jnci/dju108. J Natl Cancer Inst. 2014. PMID: 24832786 No abstract available.
References
-
- Braun S, Vogl FD, Naume B, et al. A pooled analysis of bone marrow micrometastasis in breast cancer. N Engl J Med. 2005;353(8):793–802 - PubMed
-
- Janni W, Vogl FD, Wiedswang G, et al. Persistence of disseminated tumor cells in the bone marrow of breast cancer patients predicts increased risk for relapse—a European pooled analysis. Clin Cancer Res. 2011;17(9):2967–2976 - PubMed
-
- Pantel K, Brakenhoff RH, Brandt B. Detection, clinical relevance and specific biological properties of disseminating tumor cells. Nat Rev Cancer. 2008;8(5):329–340 - PubMed
-
- Cristofanilli M, Budd GT, Ellis MJ, et al. Circulating tumor cells, disease progression, and survival in metastatic breast cancer. N Engl J Med. 2004;351(8):781–791 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

