Dissecting the transcriptional phenotype of ribosomal protein deficiency: implications for Diamond-Blackfan Anemia
- PMID: 24835311
- PMCID: PMC4058751
- DOI: 10.1016/j.gene.2014.04.077
Dissecting the transcriptional phenotype of ribosomal protein deficiency: implications for Diamond-Blackfan Anemia
Abstract
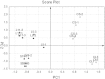
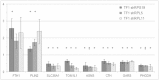
Defects in genes encoding ribosomal proteins cause Diamond Blackfan Anemia (DBA), a red cell aplasia often associated with physical abnormalities. Other bone marrow failure syndromes have been attributed to defects in ribosomal components but the link between erythropoiesis and the ribosome remains to be fully defined. Several lines of evidence suggest that defects in ribosome synthesis lead to "ribosomal stress" with p53 activation and either cell cycle arrest or induction of apoptosis. Pathways independent of p53 have also been proposed to play a role in DBA pathogenesis. We took an unbiased approach to identify p53-independent pathways activated by defects in ribosome synthesis by analyzing global gene expression in various cellular models of DBA. Ranking-Principal Component Analysis (Ranking-PCA) was applied to the identified datasets to determine whether there are common sets of genes whose expression is altered in these different cellular models. We observed consistent changes in the expression of genes involved in cellular amino acid metabolic process, negative regulation of cell proliferation and cell redox homeostasis. These data indicate that cells respond to defects in ribosome synthesis by changing the level of expression of a limited subset of genes involved in critical cellular processes. Moreover, our data support a role for p53-independent pathways in the pathophysiology of DBA.
Keywords: Bone marrow failure; Diamond Blackfan Anemia; Ribosomal protein; Ribosomopathy.
Copyright © 2014. Published by Elsevier B.V.
Figures


Similar articles
-
Ribosomal protein deficiency causes Tp53-independent erythropoiesis failure in zebrafish.Int J Biochem Cell Biol. 2014 Apr;49:1-7. doi: 10.1016/j.biocel.2014.01.006. Epub 2014 Jan 11. Int J Biochem Cell Biol. 2014. PMID: 24417973
-
Lymphoblastoid cell lines from Diamond Blackfan anaemia patients exhibit a full ribosomal stress phenotype that is rescued by gene therapy.Sci Rep. 2017 Sep 20;7(1):12010. doi: 10.1038/s41598-017-12307-5. Sci Rep. 2017. PMID: 28931864 Free PMC article.
-
Transcriptome analysis of the zebrafish model of Diamond-Blackfan anemia from RPS19 deficiency via p53-dependent and -independent pathways.PLoS One. 2013 Aug 19;8(8):e71782. doi: 10.1371/journal.pone.0071782. eCollection 2013. PLoS One. 2013. PMID: 23990987 Free PMC article.
-
Diamond Blackfan anemia: ribosomal proteins going rogue.Semin Hematol. 2011 Apr;48(2):89-96. doi: 10.1053/j.seminhematol.2011.02.005. Semin Hematol. 2011. PMID: 21435505 Review.
-
Nucleolar stress in Diamond Blackfan anemia pathophysiology.Biochim Biophys Acta. 2014 Jun;1842(6):765-8. doi: 10.1016/j.bbadis.2013.12.013. Epub 2014 Jan 8. Biochim Biophys Acta. 2014. PMID: 24412987 Review.
Cited by
-
Deficiency of ribosomal protein S26, which is mutated in a subset of patients with Diamond Blackfan anemia, impairs erythroid differentiation.Front Genet. 2022 Dec 12;13:1045236. doi: 10.3389/fgene.2022.1045236. eCollection 2022. Front Genet. 2022. PMID: 36579335 Free PMC article.
-
Ribosomopathies and the paradox of cellular hypo- to hyperproliferation.Blood. 2015 Feb 26;125(9):1377-82. doi: 10.1182/blood-2014-10-569616. Epub 2015 Jan 9. Blood. 2015. PMID: 25575543 Free PMC article.
-
Identification and fine mapping of nuclear and nucleolar localization signals within the human ribosomal protein S17.PLoS One. 2015 Apr 8;10(4):e0124396. doi: 10.1371/journal.pone.0124396. eCollection 2015. PLoS One. 2015. PMID: 25853866 Free PMC article.
-
Probing the mechanisms underlying human diseases in making ribosomes.Biochem Soc Trans. 2016 Aug 15;44(4):1035-44. doi: 10.1042/BST20160064. Biochem Soc Trans. 2016. PMID: 27528749 Free PMC article. Review.
-
Ribosomopathies: how a common root can cause a tree of pathologies.Dis Model Mech. 2015 Sep;8(9):1013-26. doi: 10.1242/dmm.020529. Dis Model Mech. 2015. PMID: 26398160 Free PMC article. Review.
References
-
- Ambekar C., Das B., Yeger H., Dror Y. SBDS-deficiency results in deregulation of reactive oxygen species leading to increased cell death and decreased cell growth. Pediatric Blood & Cancer. 2010;55:1138–1144. - PubMed
-
- Bogliolo M., Cabre O., Callen E., Castillo V., Creus A. The Fanconi anaemia genome stability and tumour suppressor network. Mutagenesis. 2002;17:529–538. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

